April 30, 2026
NEW YORK — In a major development for the treatment of blood cancer, Pfizer announced on April 29, 2026, that its immunotherapy drug Elrexfio (elranatamab-bcmm) successfully met the primary goal of its Phase 3 MagnetisMM-5 clinical trial. The study found that Elrexfio significantly extended the time patients with relapsed or refractory multiple myeloma lived without their disease worsening compared to a standard three-drug therapy.
The findings mark a critical milestone in oncology, potentially moving this advanced “bispecific antibody” from a last-resort option to a treatment used much earlier in the course of the disease. For the estimated 35,000 Americans diagnosed with multiple myeloma each year, the results offer the prospect of a more effective, non-chemotherapy alternative to manage a cancer that, while still incurable, is increasingly being treated as a chronic, manageable condition.
Moving to the Front Lines: The MagnetisMM-5 Findings
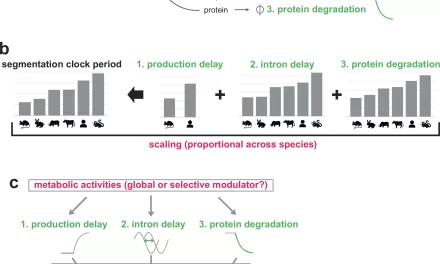
The MagnetisMM-5 study is a global, open-label trial involving nearly 500 adult patients. These participants were classified as “double-class exposed,” meaning their cancer had already stopped responding to two standard classes of treatment: proteasome inhibitors and immunomodulatory agents.
Researchers compared the effectiveness of Elrexfio as a single agent against a high-performing “triplet” regimen of daratumumab, pomalidomide, and dexamethasone.
-
Primary Success: The trial met its primary endpoint of progression-free survival (PFS). This means patients receiving Elrexfio went significantly longer before their cancer began to grow again compared to those on the standard triplet therapy.
-
Clinical Significance: While Pfizer has not yet released the exact median PFS numbers or hazard ratios, the company described the improvement as “clinically meaningful,” a term used in medicine to indicate that the benefit is substantial enough to change how doctors treat the disease.
-
Ongoing Monitoring: Data on overall survival (OS)—whether the drug actually helps patients live longer—are not yet “mature.” The trial will continue to follow participants to determine if the delay in cancer progression translates into a long-term survival advantage.
How It Works: “Bridging” the Immune System
Elrexfio belongs to a class of drugs known as bispecific antibodies. Unlike traditional chemotherapy, which kills both healthy and cancerous cells, Elrexfio acts as a molecular matchmaker.
One “arm” of the antibody attaches to the B-cell maturation antigen (BCMA) found on the surface of myeloma cells. The other “arm” grabs onto CD3, a protein on T cells (the immune system’s primary “soldiers”). By physically pulling the T cell into contact with the cancer cell, the drug directs the patient’s own immune system to recognize and destroy the malignancy.
“This data reinforces our confidence in Elrexfio’s potential to benefit patients earlier in their treatment journey,” said Dr. Jeff Legos, Pfizer’s Chief Oncology Officer. He noted that the drug’s subcutaneous (under-the-skin) injection format offers a more convenient delivery method than some other intensive immunotherapies.
Managing Risks and Side Effects
Despite the encouraging results, Elrexfio is a potent medication with a specific safety profile. The MagnetisMM-5 trial reported no new safety signals, but known risks from previous studies remain a focus for clinicians.
-
Cytokine Release Syndrome (CRS): This is a systemic inflammatory response that can cause high fevers, low blood pressure, and difficulty breathing. It is a common side effect of “re-tuning” the immune system.
-
Hematologic Toxicities: Many patients experience low white blood cell counts (neutropenia) or anemia, which can increase the risk of infections.
-
Proactive Care: To mitigate these risks, doctors use “step-up dosing”—starting with smaller priming doses—and provide patients with steroids and fever-reducing medications before treatment.
The Changing Landscape of Multiple Myeloma
Multiple myeloma accounts for roughly 10% of all blood cancers. For decades, treatment was limited to toxic chemotherapies and bone marrow transplants. The arrival of BCMA-targeted therapies like Elrexfio represents a “transformative” shift, according to independent oncologists.
However, the field is becoming increasingly crowded. Elrexfio must compete with other bispecific antibodies like teclistamab, as well as CAR-T cell therapies.
“The BCMA space is transformative but crowded,” noted experts in the field. “Each new agent must be judged not only by its efficacy but also by its toxicity profile, convenience, and real-world accessibility.”
For public health, the shift toward earlier use of targeted agents could reduce the need for more grueling therapies later on. However, the high cost of these “biologic” drugs remains a significant hurdle for global health equity and patient access.
What This Means for Patients
For now, the standard of care for myeloma patients remains unchanged until regulatory agencies, such as the FDA, review the new data and potentially expand Elrexfio’s approved use. Currently, the drug is only approved for patients who have failed at least four prior lines of therapy.
Practical takeaways for readers:
-
Earlier Intervention: If approved for earlier use, patients may be able to access this immunotherapy before their immune systems are weakened by numerous rounds of other treatments.
-
Consult Your Specialist: Patients with relapsed myeloma should discuss with their hematologist whether they might be candidates for bispecific antibodies or if participating in a clinical trial is appropriate.
-
The Trend Toward Immunotherapy: The success of Elrexfio highlights a broader trend in medicine where the focus is shifting away from “killing cells” and toward “teaching the immune system” to manage cancer as a chronic condition.
As research continues, the goal for many in the medical community is to turn multiple myeloma from a fatal diagnosis into a disease that patients can live with for decades, maintaining a high quality of life throughout.
References
-
Reuters. “Pfizer’s blood-cancer drug meets main goal in late-stage trial.” Reuters, April 29, 2026.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.