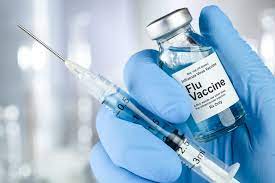
Zydus Lifesciences has launched VaxiFlu, India’s first trivalent influenza vaccine, aligning with the World Health Organization’s (WHO) 2025–26 global recommendations for seasonal flu vaccination. This vaccine targets three key influenza strains, representing a shift from previous quadrivalent flu vaccines by omitting the B/Yamagata strain, which has not circulated globally for over five years. VaxiFlu is approved for individuals aged six months and above and is expected to bolster India’s flu prevention efforts, particularly protecting vulnerable groups from severe seasonal influenza illness and deaths.
Key Developments in Influenza Vaccination in India
Zydus Lifesciences, a major Indian pharmaceutical company, introduced its trivalent influenza vaccine named VaxiFlu in early September 2025. This trivalent vaccine targets two influenza A strains (H1N1 and H3N2) and one influenza B strain (B/Victoria lineage), in line with the WHO’s updated seasonal recommendations for the Northern Hemisphere flu season 2025–26. The vaccine excludes the B/Yamagata lineage, removed from formulations globally due to no reported circulation since March 2020. Countries like the United States and nearly 40 others have already transitioned to trivalent flu vaccines following this scientific guidance.
Dr. Parvaiz Koul, a pulmonary medicine expert and fellow of the Royal College of Physicians, London, explained, “Influenza vaccination remains the most effective preventive measure against seasonal influenza and its complications. Given the global pattern of influenza virus circulation since 2020, with no circulation of influenza B Yamagata virus, regulatory bodies recommend vaccines exclude this strain. Vaccinating against a virus not in circulation for five years has no scientific merit.” This remark reinforces the rationale for India adopting the trivalent vaccine approach.
Scientific and Public Health Context
Seasonal influenza remains a significant global health problem, causing approximately 3-5 million severe illness cases and 290,000 to 650,000 respiratory deaths each year worldwide. The infection disproportionately impacts infants, elderly populations, and people with chronic illnesses such as diabetes, heart failure, kidney disease, and chronic obstructive pulmonary disease (COPD). Influenza viruses rapidly mutate, necessitating annual surveillance and vaccine composition updates by networks like WHO’s Global Influenza Surveillance and Response System (GISRS).
The shift from quadrivalent (four strains) to trivalent vaccines (three strains) aligns India’s vaccination strategy with emerging virological evidence and global regulatory guidance. This strategic move ensures that vaccines remain optimally targeted to circulating viral strains, thus maximizing effectiveness while avoiding unnecessary formulation components. It also underscores the importance of timely surveillance data for vaccine composition adjustments, providing high precision in protective coverage.
Expert Commentary and Industry Perspective
Zydus Lifesciences Managing Director Dr. Sharvil Patel commented, “We believe in aligning with global guidelines and enabling timely access to vaccines, as they are a critical part of preventive healthcare. We believe that this will significantly reduce vaccine-preventable diseases and related complications in high-risk groups.” His statement highlights the role of vaccination not only in individual protection but also in mitigating the broader public health burden of seasonal influenza.
Additionally, international and national health agencies such as the Centers for Disease Control and Prevention (CDC) and India’s National Centre for Disease Control (NCDC) have recommended the shift toward trivalent vaccines. This recommendation supports vaccination efforts ahead of the flu season, especially focusing on populations most vulnerable to influenza complications.
Practical Implications for Public Health and Individuals
For the general public and healthcare providers, the introduction of VaxiFlu means access to a vaccine tailored to current epidemiological trends, providing effective protection against the influenza virus strains most likely to circulate in the coming season. The vaccine is intended for widespread use starting from infants six months old to older adults, with emphasis on those with chronic health issues who face higher risks of severe illness.
Vaccine administration is typically recommended before the onset of peak flu season, maximizing immunity when the virus becomes more active. Individuals are encouraged to get vaccinated annually, as the virus evolves and vaccine formulations are updated accordingly. Trivalent vaccines like VaxiFlu help streamline vaccine production and distribution while maintaining protective efficacy.
Limitations and Balanced Considerations
While the trivalent vaccine excludes the B/Yamagata strain, some experts historically viewed quadrivalent vaccines as providing broader protection by covering an additional influenza B lineage. However, the absence of B/Yamagata detection worldwide for over five years supports the scientific consensus to remove it from vaccines, minimizing unnecessary vaccine components and potential side effects.
It is important to note that vaccination does not guarantee 100% protection against influenza but significantly reduces the risk of severe illness, hospitalization, and death. Continued surveillance and adaptability in vaccine formulation remain critical to address possible viral mutations or reemergence of suppressed strains.
Medical Disclaimer
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.