Australian researchers have unveiled a promising new treatment that could dramatically simplify life for people living with Parkinson’s disease: a once-a-week injectable gel that steadily delivers medication over seven days, potentially replacing the need for multiple daily pills.
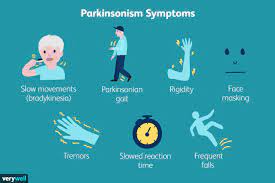
Developed by scientists at the University of South Australia (UniSA), this innovative system uses a tiny, biodegradable gel injected just under the skin or into muscle. The gel gradually releases steady doses of levodopa and carbidopa—the two main drugs used to manage Parkinson’s symptoms such as tremors, stiffness, and slowed movement. By maintaining consistent drug levels in the body, the weekly shot aims to provide steadier symptom control, reduce side effects, and greatly simplify treatment routines.
Parkinson’s disease, the world’s second most common neurological disorder, affects more than 8.5 million people globally. Current treatments often require patients—many of whom are elderly or have difficulty swallowing—to take pills several times a day. This frequent dosing can lead to fluctuating drug levels, inconsistent symptom relief, and increased risk of side effects.
Lead researcher Professor Sanjay Garg emphasized the potential impact:
“Our goal was to create a formulation that simplifies treatment, improves patient compliance, and maintains consistent therapeutic levels of medication. This weekly injection could be a game-changer for Parkinson’s care,” Prof Garg said.
The gel formulation combines an FDA-approved biodegradable polymer (PLGA) with a pH-sensitive polymer (Eudragit L-100), enabling controlled, sustained release of medication. Lab tests showed that over 90% of the levodopa and more than 81% of the carbidopa were released steadily over seven days, and the implant degraded by more than 80% within a week with no significant toxicity observed.
The injectable can be administered with a fine needle, minimizing discomfort and eliminating the need for surgical implants. Researchers are optimistic about the broader potential of this technology, which could be adapted for other chronic conditions requiring long-term drug delivery, such as diabetes or chronic pain.
The UniSA team has filed for an Australian patent and hopes to begin human clinical trials soon.
Disclaimer: This treatment is still in the experimental stage and has not yet been tested in human clinical trials. Its safety and effectiveness in patients with Parkinson’s disease have not been established. Anyone considering changes to their medical treatment should consult a qualified healthcare professional. The information provided here is for news and informational purposes only and should not be construed as medical advice.
- https://scitechdaily.com/new-once-a-week-shot-promises-life-changing-relief-for-parkinsons-patients/