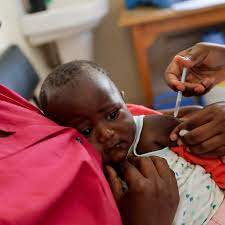
A controversial US-funded study examining hepatitis B vaccine effects on newborns in Guinea-Bissau is undergoing further ethical review by African health officials, following conflicting reports of its suspension or continuation. Health Minister Quinhi Nantote announced the pause pending technical assessment, highlighting concerns over local ethics processes after a 2025 military coup disrupted oversight. This development underscores tensions between vaccine skeptics in the Trump administration and established global health consensus on newborn immunization.
Study Background and Design
The trial, led by the Bandim Health Project—a University of Southern Denmark-affiliated group based in Guinea-Bissau—aims to enroll up to 14,000 newborns in a randomized controlled setup. Researchers plan to compare infants receiving the hepatitis B vaccine at birth against those following current local policy, which delays the first dose until six weeks alongside diphtheria-tetanus-pertussis (DTP) shots. Primary outcomes include overall mortality, hospitalizations, atopic dermatitis, and neurodevelopment over five years, building on Bandim’s prior observational work suggesting non-specific vaccine effects, such as higher female mortality post-DTP in some cohorts.
Funded with $1.6 million from the US Centers for Disease Control and Prevention (CDC) under the Trump administration—plus support from the Pershing Square and Bluebell Foundations—the study exploits a policy window before Guinea-Bissau’s planned 2027 universal birth-dose rollout. Bandim researchers argue ethical validity since no infant receives less than standard care, allowing evaluation of broader vaccine impacts beyond hepatitis B prevention. Past Bandim studies, like a 2016 analysis of 15 cohorts, reported DTP after BCG linked to 2.54 times higher mortality in girls (95% CI 1.68-3.86), fueling debates on vaccine sequencing.
Ethical Concerns Raised by Critics
Critics contend the trial withholds a proven life-saving vaccine from high-risk newborns in a country with high mother-to-child hepatitis B transmission rates, potentially leading to liver failure or cancer. Paul Offit, MD, director of the Vaccine Education Center at Children’s Hospital of Philadelphia, called it unethical: “It’s good that things are being held up while they are investigating if this is an ethical study. It’s not an ethical study.” He questioned redesign feasibility under modern standards like the Declaration of Helsinki, which mandates control groups receive the best proven methods—here, the birth dose.
Guinea-Bissau officials flagged lapses in the initial ethics review by a six-member committee that allegedly never met, exacerbated by post-coup leadership changes. Former Health Ministry official Cadija Mané emphasized poor community communication: “There is a huge lack of information about this health, or any health experiments.” The Africa CDC stepped in at Guinea-Bissau’s request for technical support, clarifying the study is “under discussion” rather than outright canceled.
Vaccine Safety and Global Recommendations
The World Health Organization (WHO) recommends hepatitis B vaccination within 24 hours of birth for all infants, regardless of maternal status, to prevent perinatal transmission—up to 95% effective when timely. In high-prevalence areas like Guinea-Bissau, the virus chronically infects many, causing 820,000 deaths yearly from liver disease. CDC and American Academy of Pediatrics echo this, advising the dose for stable newborns over 2,000 grams within 24 hours, with series completion yielding 98% immunity.
Decades of data affirm safety: no links to neurodevelopmental disorders like autism, despite study aims influenced by US Health Secretary Robert F. Kennedy Jr.’s views. Recent US policy shifts revoked universal newborn recommendations, prompting worries of rising infections.
Public Health Implications
If approved, results could inform global policies, but skeptics doubt US relevance given different epidemiology. Delays risk stalling Guinea-Bissau’s immunization plans to 2028, leaving infants vulnerable amid 1,000 annual US newborn cases despite coverage. Broader fallout includes strained US-Africa health ties, with Kennedy citing Bandim data to cut Gavi funding.
For health-conscious readers, this reinforces birth-dose importance: test pregnant women, vaccinate promptly, and complete series. In India, where hepatitis B affects millions, Universal Immunization Programme includes it at birth. (Note: Local context from general knowledge; consult NACO guidelines.)
Limitations and Balanced Perspectives
Bandim’s findings face scrutiny for survival bias and non-randomization, with WHO’s SAGE reviewing them critically. Trial power depends on enrollment before policy shift, and non-specific effects remain debated—some meta-analyses show DTP benefits. Proponents stress novelty: hepatitis B birth dose lacks large-scale overall health trials.
Diverse views include Guinea-Bissau’s sovereignty in pausing for review, prioritizing local ethics over foreign funding.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
-
Reuters. “US-funded Guinea-Bissau vaccine study to undergo ethical review, Africa CDC official says.” January 22, 2026. https://www.reuters.com/business/healthcare-pharmaceuticals/us-funded-guinea-bissau-vaccine-study-undergo-ethical-review-africa-cdc-official-2026-01-22/[reuters]