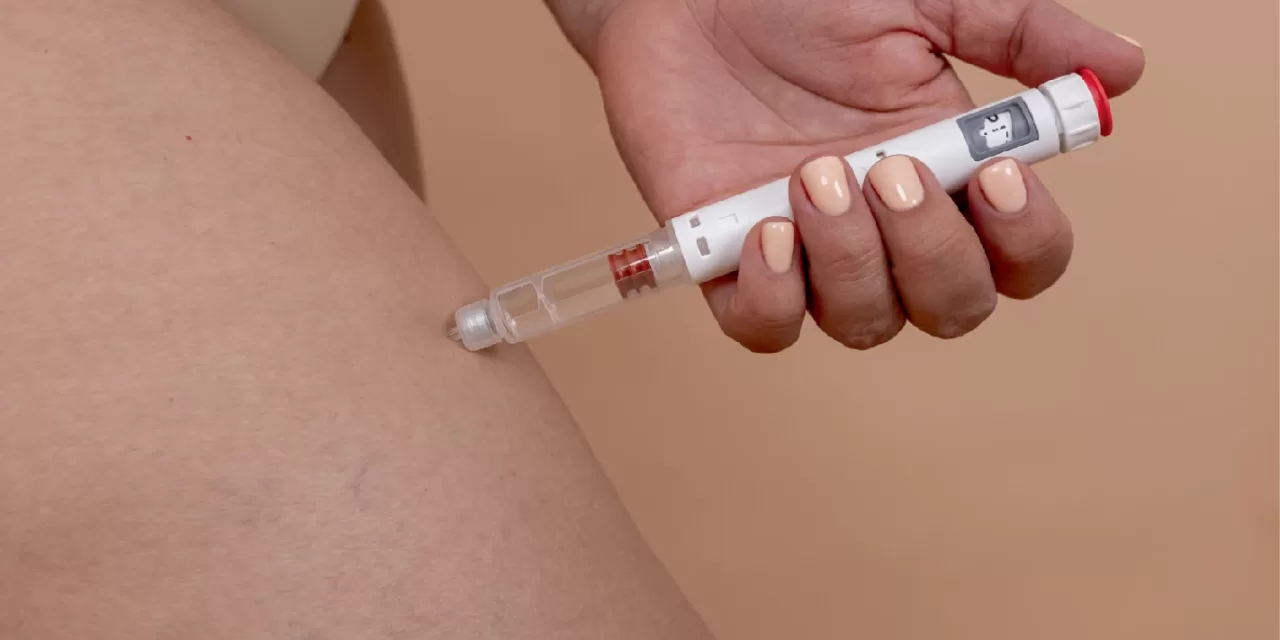
Britain’s Medicines and Healthcare products Regulatory Agency (MHRA) has approved a maximum weekly dose of 7.2 mg of semaglutide, marketed as Wegovy, for adults with obesity, marking a significant advancement in pharmacological weight management options. This decision, announced on January 16, 2026, allows eligible patients who have plateaued on the standard 2.4 mg dose to escalate treatment after at least four weeks, administered as three consecutive 2.4 mg injections. The approval stems from robust clinical evidence demonstrating superior weight loss with the higher dose when combined with diet and exercise.
Key Clinical Findings
The approval hinges on the phase 3b STEP UP trial, published in The Lancet Diabetes & Endocrinology in November 2025, involving 1,407 adults with BMI ≥30 kg/m² across 11 countries. Participants on 7.2 mg semaglutide achieved an average 18.7% body weight reduction over 72 weeks, compared to 15.6% with 2.4 mg and 3.9% with placebo; fully adherent patients saw up to 20.7% loss. Notably, 33.2% of those on the higher dose lost ≥25% of body weight, versus 16.7% on 2.4 mg and 0% on placebo, with 1.8-fold greater odds of ≥20% loss.
Dosing follows a gradual escalation: starting at 0.25 mg weekly for weeks 1-4, increasing to 0.5 mg (weeks 5-8), 1 mg (9-12), 1.7 mg (13-16), and 2.4 mg from week 17, with 7.2 mg optional from week 21 if needed. Injections for 7.2 mg must be spaced ≥5 cm apart in the same area, using multiple pens and fresh needles.
Expert Commentary
Healthcare professionals not involved in the trial emphasize the dose’s potential while urging caution. “This higher dose offers a vital tool for patients who need more support to achieve meaningful weight loss, but it must pair with lifestyle changes,” notes an endocrinologist cited in coverage of the STEP UP results. The MHRA stresses that prescribers guide usage, as the regimen targets only obesity (BMI ≥30 kg/m²), excluding overweight patients (BMI <30) or those using Wegovy for cardiovascular risk reduction.
Safety profiles mirror lower doses, with primarily mild-to-moderate gastrointestinal issues like nausea, diarrhea, and vomiting during escalation; discontinuation rates remained comparable. No new safety signals emerged, though monitoring for gallbladder issues or retinopathy in diabetics is advised.
Broader Context and Public Health Impact
Obesity affects over 26.5% of English adults, with 64.5% overweight or obese as of 2026, fueling chronic diseases and straining the NHS. Wegovy, a GLP-1 receptor agonist mimicking satiety hormones, has transformed obesity care since its UK launch, building on prior approvals up to 2.4 mg. This escalation positions the UK as the first nation to greenlight 7.2 mg, ahead of pending FDA review.
For patients, implications include enhanced options post-plateau, potentially preventing regain seen in ~20 months off-treatment per Oxford research. Public health benefits could reduce diabetes and heart disease burdens, though NICE cost-effectiveness review will determine NHS rollout.
Limitations and Balanced Perspectives
Despite efficacy, the higher dose demands three injections, potentially hindering adherence; Novo Nordisk seeks single-pen approval. Gastrointestinal side effects may intensify initially, and long-term data beyond 72 weeks is limited. Critics warn of raised expectations, with the Pharmaceutical Association noting risks if not medically supervised. Weight regain post-discontinuation underscores obesity as chronic, requiring sustained intervention.
Access barriers persist via NICE appraisal and supply issues, emphasizing holistic approaches like diet and activity.
Practical Guidance for Readers
Patients should discuss eligibility with prescribers, starting low to minimize side effects—hydrate, eat small meals, and report persistent issues. Track progress alongside BMI, waist circumference, and comorbidities. This advances personalized obesity management but reinforces multidisciplinary care.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
-
Medicines and Healthcare products Regulatory Agency (MHRA). “Medicines regulator approves up to 7.2mg dose of semaglutide (Wegovy) for patients with obesity only.” January 15, 2026. https://www.gov.uk/government/news/medicines-regulator-approves-up-to-72mg-dose-of-semaglutidewegovy-for-patients-with-obesity-o[gov]
-
Reuters. “UK health regulator approves up to 7.2 mg weekly dose of Wegovy for obesity patients.” January 16, 2026. https://www.reuters.com/business/healthcare-pharmaceuticals/uk-health-regulator-approves-up-72-mg-weekly-dose-wegovy-obesity-patients-2026-01-16/[reuters]