March 5, 2026
For decades, the “gold standard” of cervical cancer prevention has required a trip to the doctor’s office and the often-dreaded speculum exam. However, a significant shift in federal guidelines is bringing the screening process into the privacy of the home.
Updated Health Resources and Services Administration (HRSA) guidelines now officially include an option for women to self-collect samples at home for high-risk human papillomavirus (hrHPV) testing. While clinicians hail this as a “game changer” for reaching underscreened populations, the transition to home-based testing brings new complexities regarding follow-up care, insurance coverage, and the critical differences between a home swab and a traditional Pap test.
A New Era of Access
The primary goal of the updated guidelines is to close the gap in cervical cancer screening. Despite the availability of effective tests, thousands of cases of cervical cancer are diagnosed each year in the U.S., often among individuals who have not been screened in over five years.
“The big promise for at-home self-collection is to target people who would otherwise not be screened,” says George F. Sawaya, MD, director of the cervical dysplasia clinic at Zuckerberg San Francisco General Hospital. “This provides access to people who, for various reasons, don’t have access to a high-quality screening program.”
The new option is particularly significant for:
-
Individuals in rural areas with limited transportation.
-
Uninsured or underinsured patients who rely on safety-net health systems.
-
Survivors of trauma or abuse who may find traditional pelvic exams distressing.
-
Busy professionals or caregivers who struggle with scheduling in-person appointments.
Merry Jennifer Markham, MD, a gynecologic oncologist at Moffitt Cancer Center, notes that many of her patients arrive with advanced disease that could have been caught earlier. “The goal would be for me to never see these patients in my office,” she says.
How the Testing Works: Home vs. Office
It is important for consumers to understand that the at-home kit is not a “home Pap smear.” A Pap test (cytology) looks for abnormal cell changes on the cervix, whereas the at-home kit tests for the presence of the virus that causes those changes.
The Technology
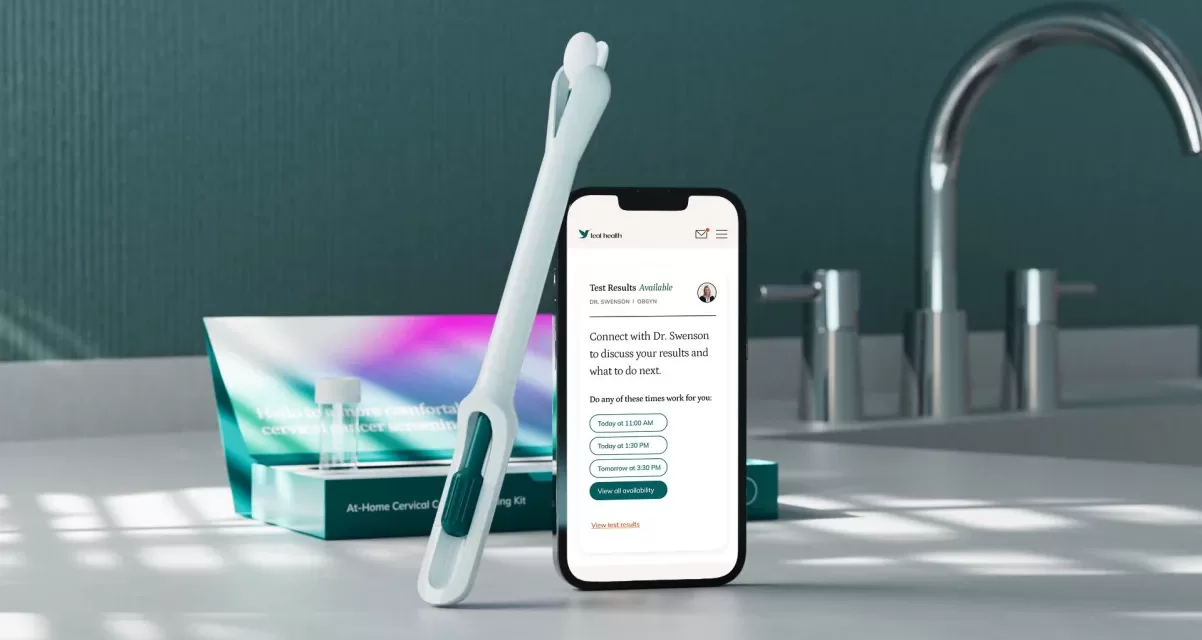
Currently, the Teal Wand is the only FDA-approved at-home self-collection device. It is used specifically with Roche’s cobas hrHPV test, a polymerase chain reaction (PCR) assay. PCR testing identifies specific DNA sequences of 14 high-risk HPV types, including HPV 16 and 18, which are responsible for about 70% of cervical cancers.
Key Procedural Differences
| Feature | At-Home Self-Collection | In-Office Clinician Collection |
| Collection Site | Vaginal wall | Cervix (via speculum) |
| Test Type | hrHPV DNA only | hrHPV and/or Cytology (Pap) |
| Sensitivity | Comparable for high-risk HPV | High |
| Follow-up | If positive, requires in-office visit | Immediate reflex testing often possible |
Understanding the Guidelines
The HRSA guidelines differ based on age and risk level. For those at average risk (asymptomatic individuals with a history of normal screenings):
-
Aged 21–29: Screening should still be done via a Pap test every 3 years. At-home HPV testing is not recommended for this age group, as HPV infections are often transient and self-clearing in younger people.
-
Aged 30–65: Primary hrHPV testing every 5 years is the preferred method for in-office visits. However, if using the at-home self-collection option, major organizations like the American Cancer Society (ACS) recommend a shorter 3-year interval.
Who Should Skip the Home Kit?
Self-collection is not for everyone. You should continue with traditional in-office clinician screening if you:
-
Are immunocompromised (e.g., living with HIV).
-
Have a history of high-grade cervical disease or cancer.
-
Were exposed to diethylstilbestrol (DES) in utero.
-
Are currently experiencing symptoms like abnormal bleeding.
The “Safety Net” of Follow-Up
A positive result on an at-home HPV test is not a cancer diagnosis; rather, it is a signal that further evaluation is needed.
“Screening is not considered complete without this follow-up,” warns Eve Rittenberg, MD, a women’s health specialist at Brigham and Women’s Hospital. If a home test comes back positive for high-risk HPV, the patient must visit a clinician for a cervical sample (cytology) or a colposcopy (a procedure to look closely at the cervix).
Experts worry that if the “loop” isn’t closed—meaning the patient doesn’t go in for that second step—the benefits of home testing vanish. Dr. Rittenberg emphasizes the need for robust tracking systems to ensure no one falls through the cracks.
Costs and Reliability
Is a home swab as accurate as a doctor’s? Evidence suggests yes. A 2018 meta-analysis published in The Lancet Oncology (Arbyn et al.) found that PCR-based HPV testing on self-samples was as sensitive as clinician-collected samples for detecting precancerous lesions.
However, financial barriers remain. The Teal Wand kit currently costs approximately $99 for those with in-network insurance and up to $249 for self-pay. While insurance coverage is expanding, it is not yet universal, which may limit the very “access” the guidelines aim to improve.
The Bottom Line
At-home HPV testing represents a major step toward patient-centered care. By removing the physical and psychological barriers of the exam room, the medical community hopes to eliminate cervical cancer as a public health threat.
“Allowing individuals the option of choosing… is a patient-centered approach that respects people’s ability to choose the strategy that is right for them,” says Dr. Rittenberg.
References
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.