WASHINGTON — In a discovery that redraws the biological map between two of humanity’s most feared diseases, researchers have identified a single protein as a master regulator of DNA repair that, when gone awry, can trigger both the nerve cell death seen in ALS and the rapid mutation characteristic of cancer.
The study, led by scientists at the Houston Methodist Research Institute and published in Nucleic Acids Research, reveals that TDP-43—a protein long known as the “hallmark” of Amyotrophic Lateral Sclerosis (ALS)—is responsible for maintaining the high-fidelity “spell-check” system of our genetic code. When this protein malfunctions, it doesn’t just stop working; it turns a vital cellular defense into a destructive force.
The “Double-Edged Sword” of DNA Repair
Every time a cell functions or divides, errors can creep into its DNA. To combat this, the body uses a process called DNA mismatch repair (MMR). Think of MMR as an elite editorial team that scans the genetic manuscript, snipping out typos (mismatched bases) and replacing them with the correct letters.
The research team, headed by Muralidhar L. Hegde, PhD, a professor of neurosurgery at Houston Methodist, found that TDP-43 acts as the supervisor for this editorial team. It specifically regulates key MMR genes like MLH1 and MSH6.
However, the study found that this relationship is precariously balanced:
-
Too Little TDP-43: In diseases like ALS and Frontotemporal Dementia (FTD), TDP-43 clears out of the cell nucleus and clumps together in the cytoplasm. This depletion causes the MMR machinery to become “hyperactive.”
-
The Result: In non-dividing neurons (which shouldn’t have much MMR activity), this overzealous repair starts “over-editing,” leading to toxic DNA breaks that kill the neuron.
-
Too Much TDP-43: Conversely, in many cancers, TDP-43 levels are abnormally high. This correlates with an increased “mutation burden,” suggesting the protein may be helping cancer cells survive or evolve by manipulating genetic stability.
“DNA repair is one of the most fundamental processes in biology,” Dr. Hegde stated. “What we found is that TDP-43 is not just another RNA-binding protein… it is a critical regulator of mismatch repair machinery. That has major implications for diseases like ALS and FTD where this protein goes awry.”
Bridging Two Medical Silos
For decades, neurodegeneration and oncology were treated as separate worlds. This discovery suggests they may be two sides of the same coin: a failure in genome guardianship.
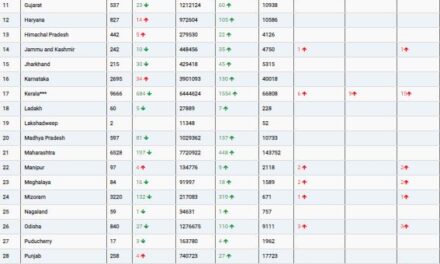
In the United States, ALS affects approximately 5 out of every 100,000 people annually. It is a devastating condition where the death of motor neurons leads to progressive paralysis. Meanwhile, cancer remains the second leading cause of death worldwide, driven by the very genetic instability that TDP-43 appears to influence.
Expert Commentary
Outside experts suggest this “cross-talk” between disciplines is the future of medicine. Dr. Aaron D. Gitler, a professor of genetics at Stanford University School of Medicine, noted that this research “reframes these diseases not as isolated silos but as interconnected failures in genome guardianship.”
Dr. Rita Satchi-Fainaro, a cancer biologist at Tel Aviv University, agreed but urged caution. “The cancer link via mutation load is compelling—high TDP-43 in tumors suggests it could be a biomarker or target, but we need clinical validation beyond databases,” she noted, emphasizing that while the data is strong, human clinical trials are the necessary next step.
What This Means for Patients and Public Health
While a “cure” is not yet on the shelf, this breakthrough provides a specific target for future drug development.
-
Precision Medicine: In lab models, researchers were able to “dial back” the excessive MMR activity, which partially reversed the damage to the cells. This hints at a future where drugs could fine-tune MLH1 or MSH6 levels to protect the brains of ALS patients.
-
Early Detection: TDP-43 could potentially serve as a “biomarker.” By monitoring the stability of a patient’s DNA or the levels of this protein, doctors might one day identify risks for ALS or certain cancers much earlier than is currently possible.
-
Genetic Counseling: For the 10-20% of ALS cases that are familial (inherited), this discovery underscores the importance of genetic screening for TDP-43 mutations, which could inform family planning and long-term health monitoring.
Limitations and the Path Ahead
As with all major breakthroughs, there are significant caveats. The study relied heavily on cell lines, animal models, and large-scale data analysis from cancer databases.
-
Causality vs. Correlation: Critics point out that high levels of TDP-43 in tumors might be a result of the cancer rather than the cause.
-
Complexity of ALS: Not all ALS patients show the same TDP-43 patterns, and the human brain is far more complex than the mouse models used in the lab.
-
The “Lynch Syndrome” Paradox: In some cases, defects in MMR cause hereditary cancers (like Lynch Syndrome), whereas this study suggests overactivity is the problem in neurons. Reconciling these different “modes” of failure will be the focus of the next decade of research.
Despite these hurdles, the study offers a rare moment of unified hope. By identifying TDP-43 as a bridge between neurodegeneration and cancer, scientists have a new map to follow—one that may eventually lead to therapies that protect the very essence of our biological identity: our DNA.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
https://health.economictimes.indiatimes.com/news/industry/scientists-discover-als-protein-that-links-dna-repair-to-cancer-and-dementia-study/129606048?utm_source=top_story&utm_medium=homepage