Published: March 5, 2026
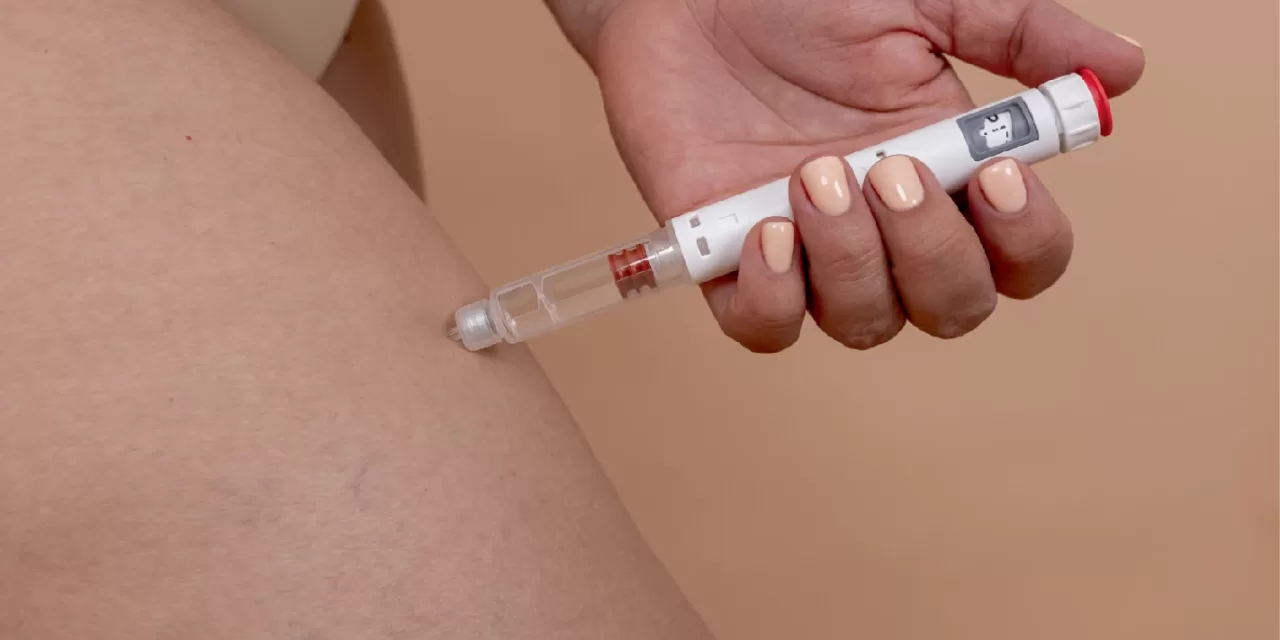
NEW DELHI — A seismic shift is hitting the Indian pharmaceutical landscape this month as the primary patents for semaglutide—the powerhouse molecule behind blockbuster drugs Ozempic, Wegovy, and Rybelsus—officially expire on March 20-21, 2026. This “patent cliff” is set to trigger an unprecedented generic onslaught, with over 50 domestic brands poised to launch more affordable versions of the GLP-1 receptor agonist. For millions of Indians living with type 2 diabetes and obesity, this development promises to slash treatment costs by 50% to 80%, transforming a high-end luxury treatment into a mainstream therapeutic option.
The Molecule That Changed the Game
Semaglutide has earned its reputation as a “game-changer” by mimicking glucagon-like peptide-1 (GLP-1), a naturally occurring hormone. It performs a triple action: stimulating insulin release, slowing gastric emptying to increase fullness, and targeting the brain’s hunger centers to curb appetite.
Clinical evidence supporting its efficacy is robust. The landmark STEP (Semaglutide Treatment Effect in People with obesity) trials, published in Nature Medicine, revealed that adults with overweight or obesity lost an average of 15-20% of their body weight over 68 to 104 weeks when using a 2.4 mg weekly dose. Beyond weight loss, participants saw significant improvements in blood pressure, waist circumference, and HbA1c levels. For those specifically managing type 2 diabetes, even lower doses (0.5–1.0 mg) resulted in sustained cardiometabolic benefits and weight reduction of nearly 7 kg over two years.
India’s “Pharmacy of the World” Steps In
While semaglutide patents in the United States and Europe are protected well into the 2030s, India’s unique patent regime—which prioritizes public health and limits “evergreening”—allows for earlier generic entry.
The Drug Controller General of India (DCGI) has already greenlit a “first wave” of domestic players. Major pharmaceutical giants are ready to flood the market with branded generics, including:
| Manufacturer | Brand Name Examples | Target Indications |
| Sun Pharma | Noveltreat, Sematrinity | Obesity & Diabetes |
| Dr. Reddy’s | Obeda, Diagluton | Obesity & Diabetes |
| Zydus Lifesciences | Mashema, Semaglyn | Obesity & Diabetes |
| Alkem Labs | Semaglide, Obesema | Multi-dose formulations |
| Torrent Pharma | Semastar, Zepsema | Broad metabolic access |
Industry analysts predict that monthly costs, which currently hover between ₹15,000 and ₹25,000 for the innovator products, could drop to as low as ₹3,500 to ₹4,000 for starter doses.
Addressing a Growing National Crisis
The timing of this generic boom is critical. According to the ICMR-INDIAB study, India is grappling with a metabolic epidemic. Estimates suggest between 101 million and 254 million people in India are living with diabetes or generalized obesity. Furthermore, the Economic Survey 2025-26 highlights a worrying trend: childhood obesity now affects 41 million individuals aged 5–19, the second-highest figure globally.
“The democratisation of GLP-1 therapies is a vital step for India,” says Dr. Arvinder Singh, a New Delhi-based endocrinologist not involved in the patent filings. “Until now, cost was the single largest barrier. However, while affordability increases access, it also increases the need for strict medical oversight to prevent misuse for ‘cosmetic’ weight loss.”
The Clinical Reality: Benefits vs. Risks
Despite the enthusiasm, medical experts urge caution. Semaglutide is a potent medication, not a “magic pill.” Data from the Mayo Clinic and JAMA Cardiology emphasize that while common side effects like nausea and vomiting are often transient, more serious risks exist.
Potential Side Effects and Limitations:
-
Gastrointestinal Issues: Nausea, diarrhea, and constipation are the most frequent complaints.
-
Rare but Serious Risks: These include pancreatitis, gallstones, and potential kidney issues. Some studies have also noted a fourfold increased risk of non-arteritic anterior ischemic optic neuropathy (a condition affecting vision) in specific populations.
-
The “Rebound” Effect: Research consistently shows that weight regain is common once the medication is discontinued, highlighting that semaglutide must be an adjunct to—not a replacement for—lifestyle modifications.
“It is a chronic medication for a chronic disease,” explains Dr. Singh. “If a patient views this as a short-term fix, they are likely to see the weight return. It requires long-term commitment to diet and exercise.”
Public Health and Economic Implications
The widespread availability of generic semaglutide could have profound economic ripple effects. Obesity-related complications are projected to cost the Indian economy ₹72 trillion by 2060. By making effective weight management more accessible, the public health system may see a reduction in the direct costs associated with treating cardiovascular disease and diabetes-related hospitalizations.
To ensure responsible use, the Central Drugs Standard Control Organisation (CDSCO) mandates that these drugs be prescribed only by registered endocrinologists or postgraduate medicine physicians. This regulatory guardrail aims to prevent the “off-label” hype that has led to global shortages and ensures that those with the highest clinical need receive priority.
Looking Ahead: The Road to March 21
As the countdown to the patent expiry begins, Indian pharma firms are scaling up complex peptide manufacturing to meet what is expected to be an “explosion” in demand. While the “slum of brands” may initially confuse consumers, the competition is expected to drive innovation in delivery mechanisms, such as more user-friendly injection pens and oral formulations.
For the Indian patient, the message is one of cautious optimism. The generic revolution offers a powerful new tool in the fight against metabolic disease, provided it is used under the guidance of healthcare professionals.
Medical Disclaimer
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
-
Medical Dialogues. (2026, March). “India’s Semaglutide Market Set for Generic Boom as Patents Expire.”