London — New groundbreaking research published in Nature Communications has uncovered the ‘roadmap’ that aggressive cancer cells follow to spread throughout the body. This discovery could change the way we approach cancer treatment by identifying metastasizing tumors earlier and preventing their spread.
The research, led by Professor Victoria Sanz Moreno and conducted by a team from The Institute of Cancer Research (ICR) London and Barts Cancer Institute at Queen Mary University of London (BCI-QMUL), marks the culmination of nearly a decade of work that began at King’s College London. The findings show how the extracellular matrix (ECM)—the scaffolding structure surrounding tumors—serves as a guide, triggering alterations in cancer cells that allow them to change shape and escape their primary location.
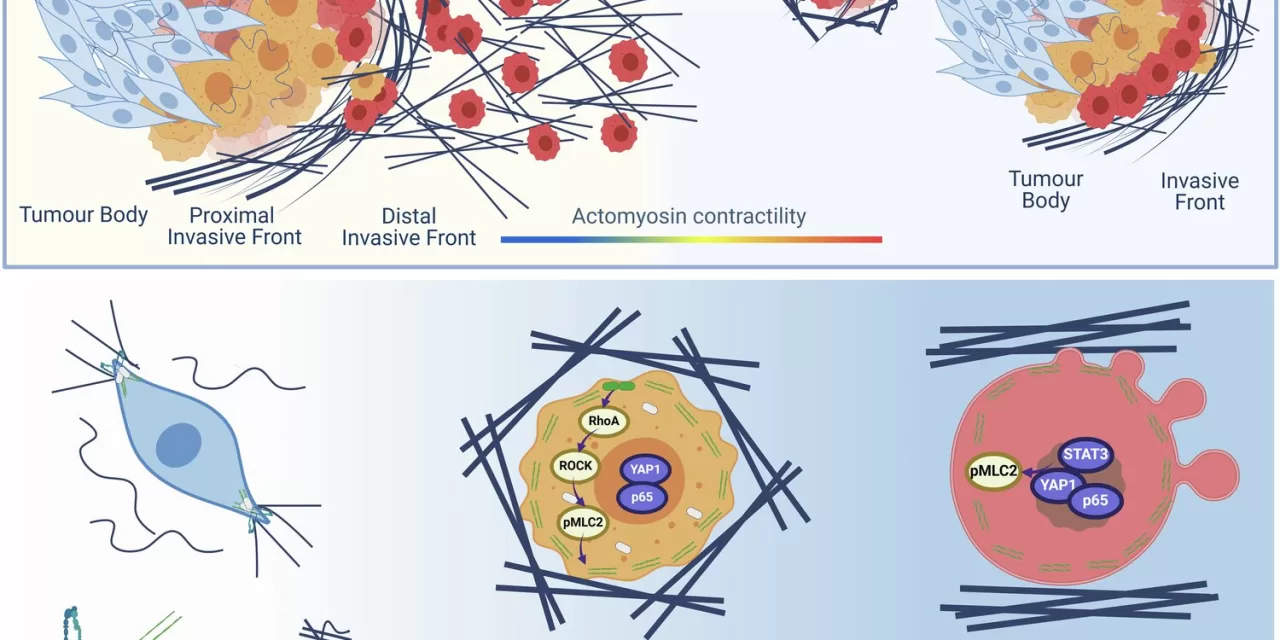
The ECM, which holds tumors together like the frame of a building under construction, has long been a topic of interest for cancer researchers. However, the new study reveals how this matrix can be reorganized in specific areas to enable cancer cells to migrate. At the tumor’s outer edges, the ECM fibers are denser and more structured, creating “tracks” that cancer cells follow as they escape and spread to other parts of the body.
By examining tumor tissue from 99 patients with melanoma and breast cancer, the team discovered that the ECM’s layout differs across the tumor. The fibers near the tumor’s center are disorganized, while those at the periphery are tightly packed and aligned in a way that directs the cancer cells outward. These changes are accompanied by cellular alterations, making the cancer cells more aggressive and capable of invading other organs.
In their experiments, the team grew melanoma cells in an ECM model resembling the conditions at the tumor’s outer edge. When injected into mice, these cells showed a greater propensity for spreading to the lungs and metastasizing than cells grown under control conditions. Additionally, the border cells exhibited an increased expression of genes associated with cell migration and inflammation, further emphasizing the aggressiveness of these cells.
The findings are significant for early detection and treatment strategies. Tumors that display these ECM characteristics are more likely to metastasize, and the researchers suggest that drugs targeting ECM components, including the enzyme lysyl oxidase (LOX), could stop the spread of cancer. LOX is already being explored in clinical trials for other conditions, and its inhibition may prevent cancer cells from using the ECM as a roadmap to invade other tissues.
Comparing the results from 14 different tumor types—including melanoma, breast cancer, pancreatic cancer, and glioblastoma—the researchers found that the presence of these metastasis-linked genes was associated with shorter survival times. This discovery not only offers a new way to predict cancer progression but also points to potential therapeutic targets to stop cancer in its tracks before it can spread.
Professor Sanz Moreno, the study’s lead researcher, expressed optimism about the future: “Now that we understand the ‘roadmap’ cancer cells follow to break out of a tumor, we can explore ways to stop this process, preventing aggressive cancers from spreading. Our research paves the way for targeting the ECM more effectively and improving the delivery of cancer treatments.”
As the fight against cancer continues, these findings offer hope that, with further research, treatments could evolve to target not just the tumor itself but the very pathways that enable its spread.
Disclaimer: The information in this article is based on research findings and should not be interpreted as medical advice. Consult with a healthcare professional for personalized guidance and treatment. The referenced study is titled “Matrix mechano-sensing at the invasive front induces a cytoskeletal and transcriptional memory supporting metastasis,” published in Nature Communications (2025).