In a significant leap for cancer research, scientists from Rutgers Health and collaborating institutions unveiled fresh findings on October 19, 2025, explaining how acute myeloid leukemia (AML) cells evade even leading treatments such as venetoclax, a breakthrough that may shape future oncology protocols worldwide. The research identifies key cellular mechanisms that allow cancer cells to cheat cell death and proposes new therapeutic angles for one of the deadliest blood cancers in adults.
Key Findings and Developments
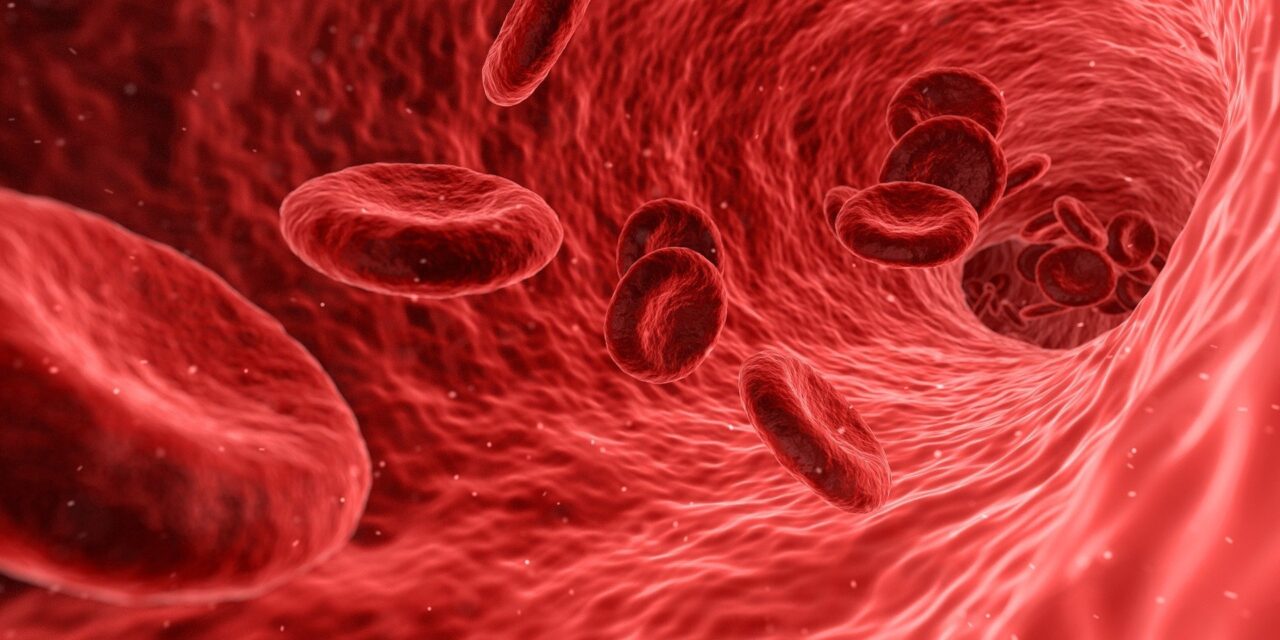
A team led by Dr. Christina Glytsou, assistant professor at Rutgers’ Ernest Mario School of Pharmacy, discovered that AML cells alter the shape of their mitochondria—the energy-producing structures within cells—to bypass apoptosis, the programmed self-destruction triggered by treatments like venetoclax. This cellular remodeling renders the standard therapy less effective over time, a cause of frequent relapse in patients. The study, published in Science Advances, demonstrated that blocking a specific protein involved in mitochondrial reshaping successfully restored drug action and improved survival in animal models.
Venetoclax induces initial remission by activating apoptosis, but most patients—about 70%—eventually develop resistance, contributing to a sobering five-year survival rate of just 30% for AML. Each year, approximately 11,000 Americans succumb to this aggressive disease.
Expert Commentary
While not involved in the research, Dr. Siddhartha Mukherjee, noted hematologist and cancer biologist, explains: “These new findings shed light on why current therapies lose ground and underscore the central role of mitochondrial dynamics in cancer resistance. Therapeutic strategies that target these energy pathways could help convert temporary cancer control into lasting remission.”
Similarly, Dr. Thoas Fioretos of Lund University notes that therapies targeting unique surface proteins or metabolic pathways in leukemia cells are emerging as promising and potentially less toxic options for patients. This aligns with the Rutgers team’s findings, highlighting energy remodeling as a new targetable vulnerability.
Context and Background
AML is marked by the rapid, uncontrolled growth of abnormal blood cells, disrupting normal blood formation. Treatment innovation has improved survival for some, especially with targeted agents like venetoclax, but resistance has remained an obstacle. The persistence of drug-resistant leukemia stem cells, shaped by genetic and epigenetic factors, frequently drives disease recurrence—even after initially promising therapy responses.
Epigenetic dysregulation, such as changes in DNA methylation and chromatin architecture, further ensures survival of AML cells under treatment, suggesting the interplay between cell metabolism and gene regulation as a research priority.
Implications for Public Health
Understanding how leukemia cells retool their energy machinery and evade apoptosis has immediate and practical implications. By blocking the protein that enables mitochondrial reshaping, scientists could potentially boost the durability of anti-leukemia drugs and reduce the risk of relapse. As noted by the Rutgers team, animal studies showed that this approach can prolong survival without notable toxicity, providing a license to pursue clinical trials.
Early interventions targeting cellular energy pathways or surface proteins might be integrated with conventional therapies, offering hope for prolonging remission, improving quality of life, and raising overall survival rates. For health-conscious readers and professionals, these developments emphasize the importance of research in disease mechanisms rather than solely drug design, supporting the move toward combination and precision medicine approaches.
Potential Limitations and Counterarguments
Despite the promise, translating these discoveries into clinical practice will require careful testing in humans. The protein-targeting compounds used were tested in animal models; differences in metabolism, toxicity, and efficacy may emerge in people. Moreover, cancer cells are notorious for developing multi-layered resistance strategies—what works today may spur new resistance tomorrow.
Other experts caution that cancer treatment advances must be viewed in the context of individual patient variability—factors like comorbidities, genetic mutations, and previous treatment history will shape real-world outcomes. Still, the reversibility of epigenetic and metabolic adaptations means these pathways remain attractive for ongoing research.
Practical Implications
For patients and families, these findings underscore the value of ongoing participation in clinical trials and open communication with oncology care teams about emerging therapy options. Enhanced understanding of resistance can help clinicians select treatment combinations or experimental therapies that are more likely to succeed. For the broader public, this research highlights the necessity of sustained investment in cancer research and patient education.
Medical Disclaimer
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References