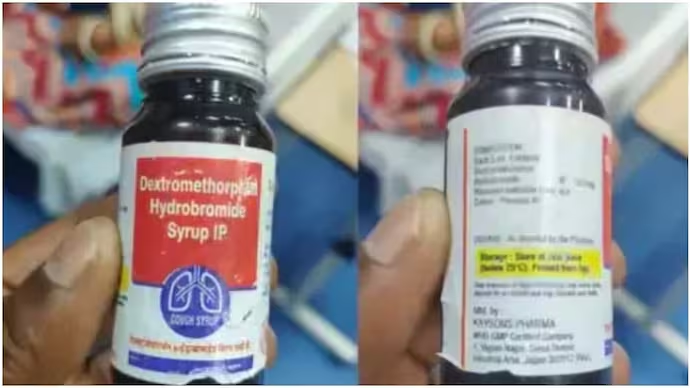
In a decisive response to the tragic deaths of children believed to be caused by substandard cough syrups, the Rajasthan government has suspended the state’s Drug Controller, Rajaram Sharma, and imposed a ban on 19 medicines supplied by Kayson Pharma, the key pharmaceutical company implicated in the controversy. This crackdown follows the deaths of at least two children and illnesses in several others after consuming government-distributed cough syrups containing the active ingredient dextromethorphan. The developments signal a stringent regulatory shift aimed at safeguarding public health and restoring trust in essential medicines dispensed under the Chief Minister’s Free Medicine Scheme.
Key Developments and Government Action
On October 3, 2025, Rajasthan authorities announced the suspension of Drug Controller Rajaram Sharma due to alleged negligence and interference in quality control processes, including tampering with medicine standards based on salt content. Concurrently, distribution of all 19 medicines produced by Kayson Pharma, a Jaipur-based pharmaceutical firm, was halted following evidence of quality control failures. These medicines included dextromethorphan-based cough syrups that have been linked to the deaths of children in the state.
The Rajasthan Medical Services Corporation Limited (RMSCL) reported that since 2012, out of over 10,000 samples tested from Kayson Pharma products, 42 were found to be substandard. In response to this public health emergency, Chief Minister Bhajan Lal Sharma has ordered a detailed investigation, forming an expert committee to meticulously review how these medicines passed quality assurance checks despite apparent lapses. A separate committee is tasked with recommending further actions in the public interest.
Banning Dextromethorphan Among Children
The state government has specifically banned all cough syrups containing dextromethorphan from all manufacturers after several batches failed quality tests. Health advisories now caution strongly against the use of dextromethorphan in children under two years of age, following guidance from the central health authorities. Additionally, mandatory labeling is being introduced to warn consumers about risks for children and pregnant women.
Medical experts caution that while dextromethorphan is widely used to suppress coughs, its safety profile in young children is controversial. The drug is generally not recommended for children below four years, and administration in younger age groups should be strictly supervised by healthcare professionals. The deaths in Rajasthan highlight the risks posed by improper use and potential quality issues with medicines dispensed through public health programs.
Context and Background
This tragic incident echoes earlier global and Indian episodes where contaminated or substandard cough syrups caused child fatalities. In 2022, over 60 children died in The Gambia following consumption of contaminated Indian-manufactured cough syrups containing toxic chemicals like diethylene glycol. The World Health Organization (WHO) issued alerts, prompting India to strengthen regulatory oversight, including the banning of unapproved fixed-dose combinations and mandatory batch testing of exported syrups. Despite these measures, lapses continue to occur in pharmaceutical quality control.
In Rajasthan, deaths were reported after children received cough syrups under a free medicine scheme at government health centers. For example, a five-year-old boy in Sikar district died hours after receiving syrup containing dextromethorphan hydrobromide. An investigating team from the Indian Council of Medical Research (ICMR) collected samples from affected areas, sending them to the National Institute of Virology for analysis. Notably, tests found no presence of known toxic contaminants like diethylene glycol or ethylene glycol in these samples, indicating the problem likely lies in inappropriate use or formulation issues rather than external contamination.
Implications for Public Health
The crisis spotlights the critical need for stringent drug quality oversight in government health programs, especially for pediatric medications. It raises questions about the rational use of cough syrups in children, who generally have self-limiting respiratory illnesses that do not require aggressive pharmaceutical treatment. Health authorities are now advocating alternative, safer approaches to managing cough symptoms in children, emphasizing careful assessment and avoiding unnecessary medication.
Moreover, this case underscores the importance of transparent regulatory mechanisms and accountability to prevent compromised products from reaching vulnerable populations. Mandatory warnings and restrictions on sales and prescriptions for cough syrups containing potentially harmful ingredients are essential steps to protect child health.
Expert Perspectives
Dr. Anjali Mehta, a pediatrician not involved in the investigation, commented, “Cough syrups containing dextromethorphan carry risks in young children and should be used judiciously. Parents and caregivers must consult qualified healthcare providers before administering such medicines. This tragedy in Rajasthan highlights gaps in both drug quality control and public education about appropriate medication use.”
Dr. P.K. Singh, a pharmacologist, added, “The suspension of the drug controller and the ban on Kayson Pharma medicines are necessary corrective actions. However, the state and national drug regulatory bodies need to improve routine surveillance and enforcement to prevent such incidents. Quality assurance cannot be compromised when children’s lives are at stake.”
Potential Limitations and Counterarguments
While the recent samples tested did not contain toxic contaminants commonly associated with cough syrup poisoning, the cause of death may also involve overdose or adverse drug reactions in susceptible children. Some experts argue that rather than banning specific medications, broader community education about the limited role of cough syrups in pediatric care could reduce harm. Others caution that removing access to symptomatic relief might lead to inappropriate alternatives, underscoring the need for balanced clinical guidelines.
Medical Disclaimer
This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to treatment plans. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
-
Rajasthan suspends drug controller, bans 19 medicines by Kayson Pharma after child deaths. India Today.