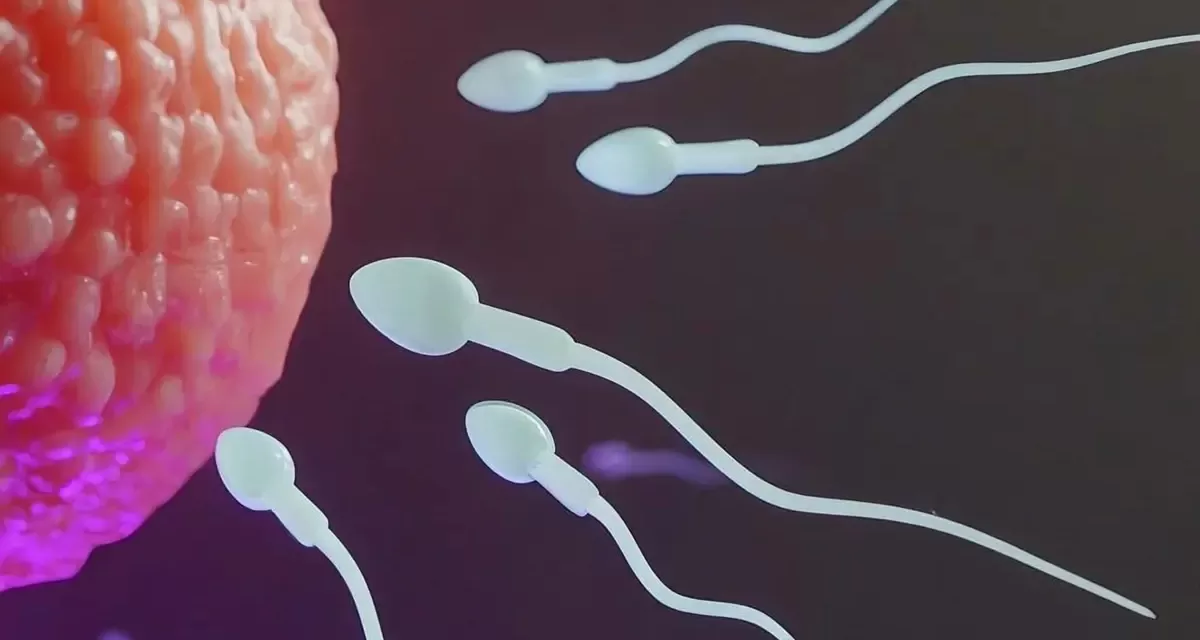
NEW YORK/MINNEAPOLIS – In a significant step towards expanding contraceptive options, scientists have begun the second phase of human trials for a novel, non-hormonal male birth control pill known as YCT-529. This development offers hope for a long-awaited alternative for men, potentially balancing the contraceptive burden currently carried primarily by women.
Developed through a collaboration between the University of Minnesota, Columbia University, and the company YourChoice Therapeutics, YCT-529 works by preventing sperm production. Crucially, it achieves this without interfering with testosterone levels, meaning it is not expected to impact the user’s libido – a common concern with potential hormonal methods.
The move to Phase 2 human trials follows encouraging preclinical results published in the journal Communications Medicine in March. Studies conducted on rats and primates demonstrated the pill’s high efficacy, proving 99% effective in preventing pregnancy in female partners. Animal studies also showed the effect was reversible: contraception took effect within two to four weeks (depending on the species), and fertility returned within six to fifteen weeks after stopping the treatment.
The initial phase of human testing (Phase 1), focused on ensuring the drug’s safety in a small group of participants, has been successfully completed. Now, a larger group of volunteers in New Zealand is participating in Phase 2 trials designed to rigorously evaluate whether YCT-529 is effective in preventing sperm production in humans. Researchers anticipate initial results from this crucial phase later this year (2025).
Currently, while numerous contraceptive methods exist for women, many come with side effects and potential health risks. The availability of a safe and effective male option has been a long-standing goal in reproductive health.
“A safe and effective male contraceptive will provide more fertility control options for couples,” stated researcher Gunda Georg of the University of Minnesota in a press release. “It will also enable more equal accountability in family planning and reproductive autonomy for men.”
The successful development and approval of YCT-529 could represent a major breakthrough, offering men a reversible, non-hormonal method to actively participate in family planning.
Disclaimer: This news article is based on information provided regarding the YCT-529 clinical trials as of April 22, 2025. The development of new medications involves extensive testing and regulatory review. The final availability, safety profile, and efficacy of this potential male contraceptive are dependent on the successful completion of all clinical trial phases and subsequent approvals by health authorities.
https://www.metropoles.com/saude/anticoncepcional-masculino-teste-humanos