Glucagon-like peptide-1 receptor agonists (GLP-1 RAs), such as semaglutide and tirzepatide—widely prescribed for type 2 diabetes management and weight loss—have recently been associated with rare but serious eye health complications, according to emerging studies published in 2024 and 2025. While these medications offer transformative benefits in blood sugar control and weight reduction, new evidence suggests a small increased risk of vision problems, including damage to the optic nerve and diabetic retinopathy progression. Health experts emphasize that these risks are uncommon but warrant awareness and regular eye monitoring.
Understanding the Key Findings
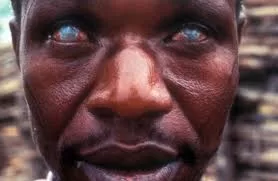
Two large observational studies involving tens of thousands of patients with type 2 diabetes encountered a modest increase in vision-related side effects among those using GLP-1 RAs. A matched cohort study including over 16,000 patients reported a doubled risk of non-arteritic anterior ischemic optic neuropathy (NAION), a condition characterized by sudden, painless vision loss caused by compromised blood flow to the optic nerve. NAION can lead to permanent vision impairment and was observed in approximately 1 in 10,000 semaglutide users, a rare incidence but with significant consequences for affected individuals.
Additionally, the same class of drugs was linked with a slight uptick in new diabetic retinopathy cases—a disease damaging the retina due to high blood sugar levels—but interestingly, the progression to severe complications like vitreous hemorrhage or blindness occurred less frequently in GLP-1 RA users compared to patients on other diabetes treatments. A study analyzing over 185,000 adults found a 24% reduction in overall blindness risk among GLP-1 users, highlighting a nuanced and complex ocular safety profile.
Expert Perspectives
Dr. Andrew Morgenstern, O.D., director of clinical resources at the American Optometric Association, underscores that “these medications are outstanding and help many people achieve diabetes control and weight loss.” He cautions that “although ocular complications are rare, if they occur, they are serious and demand prompt attention”.
Nazanin Barzideh, M.D., a retina specialist at Mount Sinai, points out that blurred vision is a common, often temporary side effect related to blood sugar fluctuations after starting GLP-1 drugs. Regarding more severe outcomes, she notes, “There is no direct evidence that these drugs cause blindness, but certain optic nerve conditions linked to GLP-1 use require vigilance and regular ophthalmologic exams,” especially within the first year of treatment.
Context and Medical Background
GLP-1 receptor agonists function by mimicking a natural hormone that increases insulin secretion and reduces appetite. They have become widely prescribed because they effectively improve blood glucose levels and contribute to substantial weight loss in patients with metabolic conditions.
NAION is a rare ocular event often described as an “eye stroke” and results from insufficient blood flow to the optic nerve. Risk factors include small optic nerve anatomy, diabetes, hypertension, and other vascular diseases—many of which coexist in patients prescribed GLP-1 drugs. Researchers hypothesize that rapid blood sugar lowering may transiently alter blood vessel function in the eye, contributing to these complications, though precise mechanisms remain unclear.
Diabetic retinopathy involves damage to retinal blood vessels stemming from chronic high blood sugar and is a leading cause of blindness in adults with diabetes. Some GLP-1 drugs have been associated with a slight increase in early retinopathy signs but also with a decrease in severe vision-threatening complications, suggesting that while new mild cases may emerge, the drugs might protect against progression to advanced eye disease.
Implications for Public Health and Patient Care
Millions of Americans use GLP-1 receptor agonists, making understanding their full safety profile essential. The overall risk of serious vision loss is low, but given the severity of potential outcomes, healthcare providers should counsel patients about warning signs such as sudden vision changes and encourage regular eye examinations.
For patients, this means that while GLP-1 drugs remain a highly effective option for diabetes and weight management, vigilance is crucial. Those with pre-existing eye disease or vascular risk factors should have scheduled screenings with eye care professionals. Prompt reporting of any visual disturbances can lead to early intervention that may preserve sight.
Limitations and Balanced Reporting
These findings arise from observational data, which cannot definitively establish causal relationships, and the absolute risk of serious eye events remains small. Confounding factors such as differences in healthcare access and patient comorbidities may influence results. Furthermore, some studies suggest potential protective effects of GLP-1 drugs against severe vision loss, highlighting the complexity of their impact on eye health.
Ongoing research is needed to clarify mechanisms, delineate risk profiles, and establish guidelines for monitoring and management. Patients and clinicians should balance the substantial benefits of GLP-1 therapies with awareness of rare ocular risks.
Medical Disclaimer
This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.