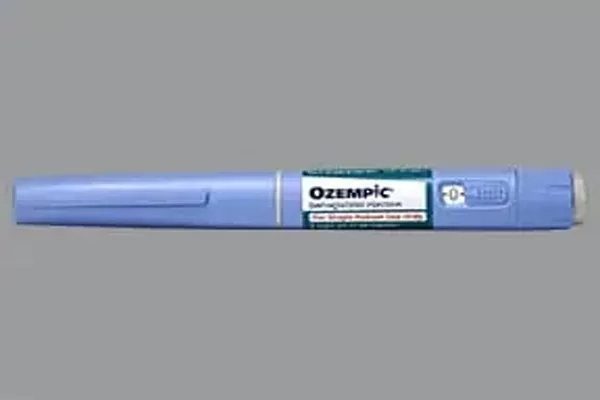
MUMBAI — Danish pharmaceutical giant Novo Nordisk is set to launch its flagship type 2 diabetes medication, Ozempic (semaglutide), in the Indian market this December. The strategic move comes just months before the drug’s patent exclusivity is scheduled to expire, signaling a race to establish brand dominance in the “Diabetes Capital of the World” before generic competitors flood the market.
A Long-Awaited Arrival
After months of regulatory anticipation, Novo Nordisk confirmed that Ozempic will be available to Indian patients starting December 2025. The drug, which has become a global cultural phenomenon for its weight-loss side effects, is officially approved in India for the treatment of type 2 diabetes mellitus in adults.
The launch follows the company’s introduction of Wegovy—a higher-dose formulation of semaglutide specifically approved for weight management—in June 2025. While Wegovy targets obesity, Ozempic is strictly indicated for glycemic control, though its off-label use for weight loss remains a significant topic of discussion in medical circles.
The “Diabetes Capital” Context
India’s diabetic population is estimated to exceed 101 million, with another 136 million classified as pre-diabetic, according to the Indian Council of Medical Research (ICMR). This staggering disease burden makes India a critical market for next-generation therapies like Glucagon-Like Peptide-1 (GLP-1) receptor agonists.
“The arrival of Ozempic offers a powerful new tool for endocrinologists in India,” says Dr. Anjali Mehta, a senior endocrinologist at a leading Mumbai hospital. “For patients with uncontrolled type 2 diabetes, particularly those with high cardiovascular risk, GLP-1 agonists have revolutionized care. However, accessibility remains the primary hurdle.”
Cost and Accessibility Barriers
Industry sources estimate the monthly cost of Ozempic in India will range between ₹10,000 and ₹20,000 (approx. $115-$230), depending on the dosage. In a country where out-of-pocket health expenditure is high and insurance coverage for chronic outpatient care is limited, this pricing places the drug in the premium segment.
Vikrant Shrotriya, Managing Director of Novo Nordisk India, emphasized the company’s commitment to availability in recent statements, noting that they are “pushing for an accelerated rollout” to serve patient needs despite the complex logistics of cold-chain supply.
The Patent Cliff and Generic Competition
The timing of this launch is critical. Novo Nordisk’s patent for semaglutide in India is set to expire in March 2026. This impending “patent cliff” has triggered a flurry of activity among domestic pharmaceutical giants.
Just days prior to the launch announcement, the Delhi High Court delivered a significant ruling allowing Dr. Reddy’s Laboratories to manufacture and export semaglutide, though it barred domestic sales until the March 2026 patent expiry. This legal victory sets the stage for a wave of affordable generic versions—likely priced 60-70% lower than the innovator drug—to enter the market by mid-2026.
“Novo Nordisk is effectively trying to capture the brand loyalty of high-income patients before Indian generics democratize access,” explains pharmaceutical analyst Rajesh Kumar. “Once the generics arrive, the market dynamics will shift drastically from a low-volume, high-margin model to a high-volume game.”
Clinical Context: How It Works
Ozempic works by mimicking the GLP-1 hormone, which prompts the body to produce more insulin when blood sugar levels rise. It also slows gastric emptying, making patients feel full longer.
Key Medical Considerations:
-
Efficacy: Clinical trials show significant reductions in HbA1c levels (a measure of long-term blood sugar).
-
Cardiovascular Benefit: The drug has demonstrated benefits in reducing the risk of major adverse cardiovascular events in diabetic patients with established heart disease.
-
Side Effects: The most common adverse effects are gastrointestinal, including nausea, vomiting, and diarrhea. Rare but serious risks include pancreatitis and thyroid C-cell tumors (observed in rodent studies).
Implications for Public Health
The launch complicates an already heated market. Eli Lilly’s rival drug, Mounjaro (tirzepatide), which targets both GLP-1 and GIP receptors, has already established a foothold in India, reportedly becoming a top-selling brand by value in October 2025.
Health experts urge caution regarding the “medicalization” of lifestyle management. “While these drugs are breakthroughs, they are not magic bullets,” warns Dr. Mehta. “They must be prescribed alongside diet and exercise changes. There is also a genuine concern about supply shortages for diabetic patients if the drug is diverted excessively for cosmetic weight loss, as seen in the US and Europe.”
Conclusion
As Ozempic hits Indian pharmacy shelves, it marks a pivotal moment in the country’s battle against diabetes. While the high cost currently limits its reach to the affluent, the looming entry of generics in 2026 promises to eventually make this life-changing therapy accessible to the millions of Indians who need it most.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References:
-
Novo Nordisk gears up for December Ozempic launch in India, Reuters (2025).
Recommended Video:
News18 Report: Novo Nordisk’s India Strategy
This video features an interview with Novo Nordisk India’s Managing Director discussing the strategic timeline for bringing Ozempic to the Indian market ahead of patent expiry.