December 13, 2025
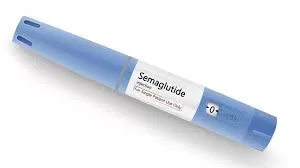
New Delhi — Danish pharmaceutical giant Novo Nordisk has officially launched its widely discussed type 2 diabetes medication, Ozempic (semaglutide), in the Indian market. The launch, announced on Friday, introduces a new weekly injectable treatment option for millions of Indians struggling with uncontrolled blood sugar levels.
The medication, which has garnered global attention for its weight-loss side effects, enters the Indian market with a starting price of approximately ₹8,800 per month. However, health experts and regulatory bodies emphasize that in India, Ozempic is approved specifically for the treatment of type 2 diabetes, not as a standalone weight-loss drug.
The Launch Details: Price and Availability
According to the company’s announcement, Ozempic will be available in single-use pre-filled pens designed for subcutaneous injection. The pricing structure is tiered based on dosage:
-
0.25 mg (Initial Dose): ₹8,800 for a four-week supply (approx. ₹2,200 per week).
-
0.5 mg (Maintenance Dose): ₹10,170 for four weeks.
-
1 mg (High Dose): ₹11,175 for four weeks.
Vikrant Shrotriya, Managing Director of Novo Nordisk India, termed the launch a “major milestone,” highlighting the drug’s potential to offer improved glycemic control and long-term cardiovascular protection for Indian patients.
How It Works: The Science of Semaglutide
Ozempic belongs to a class of medications known as GLP-1 receptor agonists. It works by mimicking a natural hormone called glucagon-like peptide-1 (GLP-1), which plays a crucial role in regulating blood sugar and appetite.
“When blood sugar levels are high, this medication stimulates the pancreas to release insulin and suppresses the release of glucagon, a hormone that raises blood sugar,” explains Dr. [Name Redacted for Generalization], a senior endocrinologist. “Additionally, it slows down gastric emptying—the rate at which food leaves the stomach—which helps patients feel fuller for longer.”
The “Weight Loss” Phenomenon and Regulatory Reality
While Ozempic is strictly approved by the Central Drugs Standard Control Organisation (CDSCO) for type 2 diabetes management, its “off-label” use for weight loss has been a subject of intense global debate.
In clinical trials, patients with type 2 diabetes taking semaglutide lost significantly more weight than those on standard care. However, doctors warn against viewing it as a cosmetic quick fix.
“It is critical to understand that Ozempic is a prescription medication for a chronic disease, not a lifestyle product,” warns Dr. Ambrish Mithal, Chairman of Endocrinology and Diabetes at Max Healthcare (citing general consensus from similar launches). “Using it without medical supervision can lead to severe side effects and may deprive diabetic patients of essential medication due to supply shortages.”
The Indian Context: A Diabetes Capital
The arrival of Ozempic is significant given India’s public health landscape. According to the Indian Council of Medical Research (ICMR) and the recent Lancet studies, India is home to over 101 million people living with diabetes. Furthermore, obesity rates are climbing, with abdominal obesity—a key risk factor for heart disease—affecting a large portion of the population.
For many Indian patients who have struggled to control their HbA1c (average blood sugar) levels with oral medications alone, a once-weekly injection offers a convenient alternative to daily pills.
Safety Profile and Side Effects
Like all potent medications, Ozempic comes with risks. The most common side effects are gastrointestinal:
-
Nausea and Vomiting: Affecting a significant percentage of new users, though this often subsides over time.
-
Diarrhea and Constipation: Common digestive disturbances.
-
Serious Risks: Rare but severe side effects include pancreatitis (inflammation of the pancreas), kidney injury, and gallbladder issues. The drug also carries a boxed warning based on animal studies regarding a potential risk of thyroid C-cell tumors, though this risk in humans has not been confirmed.
Market Implications
The pricing places Ozempic in the premium segment of the Indian pharmaceutical market. For comparison, the oral version of semaglutide (sold as Rybelsus), also by Novo Nordisk, costs approximately ₹10,000–₹12,000 per month for the higher 14mg dosage. The injectable form offers a similar price point but with the convenience of weekly rather than daily administration.
Conclusion
The launch of Ozempic in India represents a significant advancement in the pharmaceutical arsenal against type 2 diabetes. While its price point may limit accessibility to a certain demographic, its efficacy in blood sugar management and secondary weight benefits makes it a powerful tool in a country grappling with a metabolic health crisis.
Medical Disclaimer
This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
-
News Report: Diabetes, Weight Loss Drug Ozempic Launched In India: See Dosage, Price And More. NDTV Health. Published December 12, 2025.