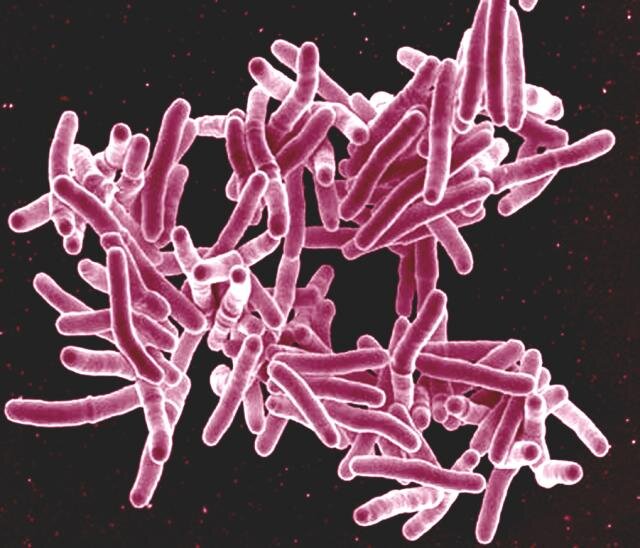
NEW YORK, NY – Tuberculosis (TB) remains a major global health threat, claiming approximately 1.25 million lives in 2023, more than any other infectious disease, despite being curable. While standard antibiotic regimens successfully cure about 88% of patients globally, a significant portion still experience treatment failure. A key challenge is the lack of a test to determine precisely when the Mycobacterium tuberculosis bacteria have been eliminated, leading to a standardized, lengthy treatment that may be too long for some and insufficient for others.
Researchers at Weill Cornell Medicine, led by Dr. Kayvan Zainabadi, assistant professor of molecular microbiology in medicine, are developing a novel RNA-based diagnostic test designed to overcome this hurdle.
“We don’t have a test to tell someone if they are cured,” stated Dr. Zainabadi. “So, right now clinicians rely on a one-size-fits-all, six-month treatment approach even though we know that 5%-10% of patients won’t be cured under this regimen. At the same time, a majority of patients will achieve cure in less than six months and thus end up taking unnecessary antibiotics.”
The proposed test targets the TB bacterium’s 16S ribosomal RNA (rRNA), a highly abundant molecule essential for protein production within the cell. Unlike bacterial DNA, which can persist in the body long after successful treatment and lead to false positives in current DNA-based tests, RNA degrades much more quickly.
“DNA from the TB bacterium can hang around in patients for months and even years after successful cure, so it’s not a good proxy for disease,” Dr. Zainabadi explained. “RNA, with its shorter half-life, is.”
This RNA-based approach aims to provide a clear signal of active infection, similar to how viral load assays monitor HIV treatment effectiveness. “What we’re trying to accomplish is akin to what the viral load assay did for HIV, where we can accurately and rapidly assess how patients with TB are responding to therapy,” Dr. Zainabadi added.
Furthermore, the abundance of rRNA offers potential for detecting TB in samples other than sputum (coughed-up mucus), the standard diagnostic sample. This is crucial for vulnerable populations like children and people with HIV, who often cannot produce adequate sputum. Current methods for these groups can be invasive (like gastric sampling in children) and less effective.
The development of this test builds on previous work by Dr. Zainabadi in Haiti, collaborating with the GHESKIO Centers and Weill Cornell Medicine’s Center for Global Health, directed by Dr. Daniel W. Fitzgerald. Initial studies involving 41 TB patients helped refine methods for extracting and detecting TB rRNA from sputum.
Now, with a $350,000 grant from RePORT International, Dr. Zainabadi’s team, including colleagues in India and Uganda, is conducting two key studies:
- RePORT India (Pune): Using archived patient samples to determine if the RNA test can accurately identify individuals who failed the standard six-month treatment.
- RePORT Uganda (Kampala): Assessing the test’s ability to diagnose TB in children, particularly exploring the use of less invasive samples like saliva, nasal swabs, urine, and stool. Sister labs are being established in both countries to enable local testing.
If successful, this RNA-based test could significantly impact TB care by:
- Allowing clinicians to personalize treatment duration, potentially shortening it for many and ensuring completion for those needing longer therapy.
- Reducing the burden of long antibiotic courses and their associated side effects.
- Minimizing the risk of developing drug-resistant TB due to incomplete treatment.
- Improving diagnostic accuracy, especially in children.
- Accelerating clinical trials for new TB drugs by providing faster indicators of treatment success.
“Our long-term goal is to develop a test that improves clinical outcomes for patients with TB,” Dr. Zainabadi concluded. Successful results from the current studies will pave the way for larger validation trials across diverse populations.
Disclaimer: This news article is based on information provided by Weill Cornell Medicine regarding ongoing research. The diagnostic test described is currently under development and investigation and is not yet approved or available for general clinical use.