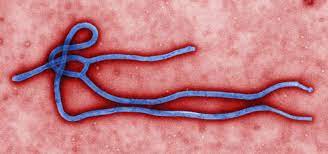
In a groundbreaking study, researchers at the La Jolla Institute for Immunology (LJI) have identified a human antibody, mAb 3A6, that could revolutionize treatment for the deadly Ebola virus. The findings, published in Nature Communications, reveal that this antibody effectively neutralizes the virus and offers protection even in the advanced stages of infection.
The antibody was discovered in blood samples from an Ebola survivor treated at Emory University Hospital during the devastating 2014-2016 outbreak, which claimed more than 11,300 lives in West Africa. By binding to a crucial viral structure known as the “stalk,” mAb 3A6 blocks infection and prevents the virus from spreading within the host.
A New Hope in Ebola Treatment
Scientists at the National Institute of Allergy and Infectious Diseases (NIAID) tested mAb 3A6 in non-human primates, with promising results. According to Dr. Erica Ollmann Saphire, President & CEO of LJI, “This antibody offers the best protection in primates at the lowest dose yet seen for any single antibody.” The study, conducted in collaboration with experts from Cambridge University, the Max Planck Institute of Biochemistry, and the Emory Vaccine Center, underscores the potential of this antibody as a cost-effective and easily manufactured therapeutic option.
Understanding How mAb 3A6 Works
The research team, led by Dr. Kathryn Hastie of LJI, used advanced imaging techniques such as cryoelectron tomography and x-ray crystallography to capture detailed visuals of how mAb 3A6 interacts with the Ebola virus. The antibody exploits the dynamic movement of viral proteins, slipping between them to secure its target and disrupt infection. “The lower the amount of an antibody you can deliver, the easier it will be to manufacture a treatment—and the lower the cost,” says Dr. Hastie.
Notably, the stalk structure targeted by mAb 3A6 is conserved across multiple Ebola virus species, making it a promising candidate for a broad-spectrum, “pan-Ebolavirus” therapeutic. Additionally, insights from this study may guide the development of vaccines designed to target this critical viral component.
Future Implications
The research was supported by several organizations, including the National Institutes of Health (NIH), the Defense Advanced Research Projects Agency (DARPA), the UK Medical Research Council, and the European Research Council. With ongoing studies and potential clinical trials on the horizon, mAb 3A6 could pave the way for more effective and accessible Ebola treatments.
Disclaimer:
This article is based on current research findings and is for informational purposes only. While promising, mAb 3A6 is still in the experimental phase and not yet approved for clinical use. Further studies and regulatory approvals are required before it can be widely available as a treatment for Ebola virus disease.