Published: January 19, 2026
PITTSBURGH — Researchers at Carnegie Mellon University (CMU) have unveiled a breakthrough molecular strategy that could transform the treatment landscape for myotonic dystrophy type 1 (DM1) and a host of other incurable neurological disorders. By designing a specialized “pothole filler” molecule that plugs genetic errors without disturbing healthy cellular machinery, the team has successfully targeted the structural root of the disease in laboratory models.
The findings, published this week in the Proceedings of the National Academy of Sciences (PNAS), offer a novel blueprint for treating “RNA-repeat expansion” disorders. These conditions—which include ALS, Huntington’s disease, and Friedreich’s ataxia—occur when the body’s genetic code gets stuck on “repeat,” creating toxic structures that hijack essential cellular functions.
The Genetic ‘Stutter’ Behind Muscle Loss
Myotonic dystrophy type 1 is the most common form of adult-onset muscular dystrophy, affecting approximately 1 in 2,300 people globally. It is characterized by progressive muscle wasting, weakness, and myotonia—an inability to relax muscles after contraction. However, the disease is systemic, often damaging the heart, lungs, and eyes.
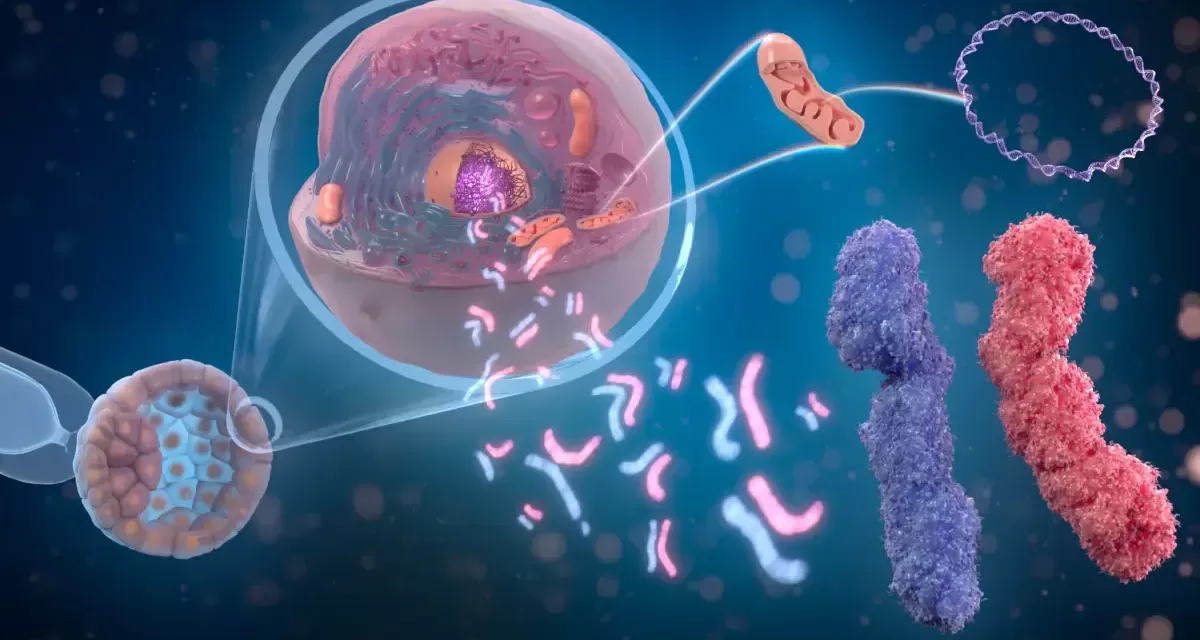
At the heart of DM1 is a mutation in the DMPK gene. In a healthy individual, a specific sequence of genetic code (CTG) repeats between 5 and 35 times. In those with DM1, this sequence can repeat thousands of times.
When the cell attempts to read this elongated instruction manual, it produces “toxic RNA.” Instead of moving through the cell smoothly, this RNA folds into a tangled, rigid structure known as a hairpin loop.
“These loops function like a sticky trap,” explains Danith Ly, Ph.D., Professor of Chemistry at CMU and Director of the Institute for Biomolecular Design and Discovery. “Essential proteins that are supposed to help the cell function get stuck in these traps. This creates a cellular traffic jam, preventing the production of other vital proteins.”
A ‘Two-Faced’ Solution to a Tangled Problem
Current experimental therapies often struggle with “off-target effects,” meaning they might accidentally disable healthy genes while trying to fix the broken ones. To solve this, Dr. Ly’s team developed a new class of synthetic molecules called gamma peptide nucleic acids (PNAs) featuring Janus bases.
Named after the two-faced Roman god, these Janus bases are double-sided. This allows the drug to slide into the center of the toxic RNA hairpin and bind to both sides of the “tangle” simultaneously.
“Think of it as a pothole filler,” says Dr. Ly. “It fits neatly into the damaged spots of the genetic road without disrupting the rest of the street.”
Unlike traditional “antisense” therapies that try to pull the RNA apart—a process that requires significant energy and can be imprecise—this new ligand, dubbed LG2b, simply occupies the space. By filling the gap, it prevents the “sticky trap” from catching the proteins the cell needs to survive.
Expert Perspectives: A Shift in Precision
While the CMU team is optimistic, the broader medical community notes that while the chemistry is brilliant, the journey to a pharmacy shelf is long.
“The specificity demonstrated in this study is a significant leap forward,” says Dr. Elena Rossi, a molecular geneticist not involved in the research. “One of the greatest hurdles in RNA therapy is ensuring the drug doesn’t interfere with the ‘normal’ version of the gene. This ‘bifacial’ recognition approach appears to solve much of that stability and selectivity issue.”
However, Dr. Rossi cautions that delivering these molecules into the deep tissues of the human body—particularly the brain and skeletal muscle—remains a formidable challenge for any RNA-based treatment.
Beyond Myotonic Dystrophy
The implications of this “pothole-filling” strategy extend far beyond DM1. Because many neurodegenerative diseases share this “repeat expansion” mechanism, the same logic could theoretically be applied to:
-
Amyotrophic Lateral Sclerosis (ALS): Specifically forms linked to the C9orf72 mutation.
-
Huntington’s Disease: Caused by CAG repeats.
-
Spinocerebellar Ataxias: A group of disorders affecting coordination and balance.
“Diseases like Huntington’s and fragile X syndrome involve the repeat of only three nucleobases,” says Dr. Ly. “It seems so simple, yet the symptoms are life-stealing. If we can stop proteins from being sequestered in these hairpins, we believe we can significantly improve patient outcomes.”
What This Means for Patients
For families living with DM1, this research represents a shift from managing symptoms to addressing the underlying genetic cause. Currently, there is no cure or disease-modifying therapy for myotonic dystrophy. Treatment is limited to physical therapy, braces, and managing heart or respiratory complications.
While the LG2b ligand has shown “remarkable selectivity” in laboratory models, it has not yet entered human clinical trials. The research team is currently optimizing the molecules to improve how well they are absorbed by cells and evaluating their safety in preclinical animal models.
Statistical Context: The Scope of the Problem
| Condition | Genetic Repeat | Est. Prevalence |
| Myotonic Dystrophy (DM1) | CTG | 1 in 2,300 |
| Huntington’s Disease | CAG | 1 in 10,000 |
| Friedreich’s Ataxia | GAA | 1 in 50,000 |
The Road Ahead
The CMU study marks a milestone in structure-based RNA therapy. By targeting the shape of the toxic RNA rather than just its sequence, researchers are opening a new front in the war against genetic disorders.
As the team refines their drug delivery methods, the goal remains clear: to turn a “cellular traffic jam” back into a clear highway for human health.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
Peer-Reviewed Study:
-
Perera, J. D. R., Thrikawala, S., Thadke, S., et al. (2026). A pothole-filling strategy for selective targeting of rCUG-repeats associated with myotonic dystrophy type 1. Proceedings of the National Academy of Sciences (PNAS). DOI: 10.1073/pnas.2507065123