May 27, 2025 – In a significant breakthrough for the treatment of high blood pressure, the largest Phase III clinical trial to date investigating a novel therapy for hypertension has reported encouraging results. The findings, announced at the 34th European Meeting on Hypertension and Cardiovascular Protection, highlight the safety and effectiveness of lorundrostat, an aldosterone synthase inhibitor, in patients with uncontrolled or resistant hypertension.
A Major Step Forward in Hypertension Care
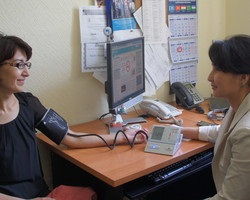
The Launch-HTN trial, led by Dr. Manish Saxena of Queen Mary University of London’s William Harvey Heart Center, enrolled a large and diverse group of patients worldwide who struggled to control their blood pressure despite taking two to five antihypertensive medications. The randomized, double-blinded, placebo-controlled study tested lorundrostat’s ability to lower blood pressure by targeting the aldosterone pathway—a key regulator in blood pressure control.
“Despite available treatments, more than 40% of adults with hypertension worldwide are not reaching their blood pressure goal,” said Dr. Saxena. “There’s a major need to explore novel therapies for hypertension and the Launch-HTN trial addressed this need.”
How Lorundrostat Works
Lorundrostat belongs to a new class of drugs called aldosterone synthase inhibitors. It works by blocking the production of aldosterone, a hormone produced by the adrenal glands that can contribute to high blood pressure and related complications such as heart failure and kidney problems. By inhibiting the enzyme CYP11B2, lorundrostat reduces aldosterone levels, helping to control blood pressure.
Trial Results: Significant and Sustained Reductions
The trial’s results were impressive:
-
At Week 6: Lorundrostat 50 mg once daily reduced systolic blood pressure by 16.9 mmHg (9.1 mmHg more than placebo).
-
At Week 12: The reduction reached 19 mmHg (11.7 mmHg more than placebo).
-
The drug demonstrated a good safety profile and was effective across various patient groups.
Potential Impact
Hypertension is a global health issue, affecting one in three adults and significantly increasing the risk of heart attack, stroke, and other cardiovascular diseases. About 30% of people with hypertension have dysregulated aldosterone, making them especially likely to benefit from treatments like lorundrostat.
Dr. Saxena emphasized, “Once commercially available, lorundrostat could be a novel treatment option for hypertension in millions of patients worldwide.”
Disclaimer:
This article is for informational purposes only and is based on preliminary results from a Phase III clinical trial as reported by MedicalXpress. Lorundrostat is not yet commercially available, and its long-term safety and effectiveness will require further evaluation and regulatory approval. Patients should not make any changes to their medication regimen without consulting their healthcare provider. For more information, visit MedicalXpress.