February 13, 2026
NEW DELHI — In a landmark finding that could redefine India’s battle against drug-resistant tuberculosis, a new economic evaluation from the Indian Council of Medical Research (ICMR) has confirmed that shorter, six-month all-oral treatment regimens are not only more effective but also significantly cheaper for the national healthcare system than current standard therapies.
The study, published this week in the Indian Journal of Medical Research, suggests that transitioning to the BPaL (Bedaquiline, Pretomanid, and Linezolid) and BPaLM (with Moxifloxacin) regimens could accelerate India’s goal of eliminating TB by optimizing resources and improving patient adherence. For a country that carries the world’s highest TB burden, these findings offer a pragmatic roadmap for scaling up high-quality care.
The Shift from Months to Weeks
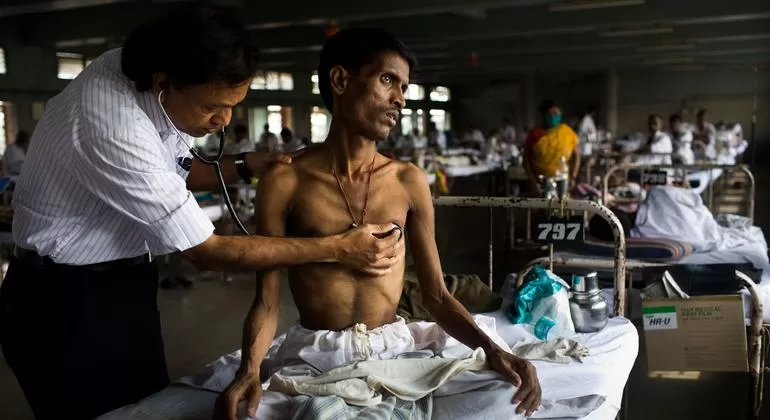
For decades, patients diagnosed with Multidrug-Resistant or Rifampicin-Resistant Tuberculosis (MDR/RR-TB) faced a grueling recovery path. Traditional treatments often lasted between 18 to 20 months, involving a cocktail of toxic drugs and, in older protocols, painful daily injections.
The new study, conducted by the ICMR–National Institute for Research in Tuberculosis (ICMR-NIRT), evaluated the cost-effectiveness of cutting that duration down to just six months using all-oral medications. Researchers compared these “shorter” regimens against the existing 9–11 month and 18–20 month protocols currently used under the National TB Elimination Programme (NTEP).
The Economic Verdict: Better Health for Less Money
The most striking finding of the report is the “win-win” nature of the BPaL regimen. In the world of health economics, a treatment is rarely both more effective and less expensive than the status quo, but BPaL achieves exactly that.
-
BPaL Regimen: The health system actually saves INR 379 per patient for every Quality Adjusted Life Year (QALY) gained compared to standard regimens.
-
BPaLM Regimen: This variation was found to be “highly cost-effective,” costing the health system a negligible INR 37 additional per patient per QALY gained.
“What we are seeing is a rare alignment of clinical excellence and fiscal responsibility,” says Dr. Ananya Sharma, a public health policy expert not involved in the study. “By reducing the treatment time from nearly two years to just six months, we reduce the burden on hospitals, the cost of long-term monitoring, and the likelihood of patients dropping out due to side effects.”
Why “All-Oral” Matters
The transition to an all-oral regimen is more than just a matter of convenience; it is a matter of human dignity and clinical success. Older TB treatments often included injectable drugs that could cause permanent hearing loss, kidney damage, and severe nausea.
“The ‘all-oral’ aspect is a game-changer for patient psychology,” explains Dr. Rajesh Varma, a pulmonologist who has treated MDR-TB for two decades. “When a patient doesn’t have to travel to a clinic for a painful injection every day, their ability to maintain a job and stay with their family improves. Adherence goes up, and the risk of developing further drug resistance goes down.”
Key Benefits Identified:
-
Faster Recovery: Patients return to their normal lives and workforce nearly a year sooner.
-
Lower Toxicity: Fewer drugs for a shorter duration typically results in fewer severe adverse events.
-
System Relief: Reduced frequency of follow-up visits and laboratory monitoring frees up resources for other primary healthcare needs.
Limitations and the Road Ahead
While the ICMR study provides a robust economic argument, experts caution that implementation comes with challenges. The BPaL regimen requires careful monitoring for specific side effects, such as peripheral neuropathy (nerve pain) or anemia, often associated with the drug Linezolid.
Furthermore, the “cost-saving” nature of the drug depends on the government’s ability to procure these newer medications—some of which are still under patent or produced by limited manufacturers—at scale.
“Economic models are vital, but the real-world success depends on the supply chain,” notes Dr. Sharma. “We must ensure that these six-month kits are available in the most remote corners of Bihar and Maharashtra, not just in urban centers.”
A National Priority
India has set an ambitious target to eliminate TB by 2025, five years ahead of the global Sustainable Development Goals. While the 2025 deadline remains a steep mountain to climb, the adoption of BPaL and BPaLM regimens under the NTEP provides the necessary leverage.
By focusing on treatments that are “person-centered”—meaning they respect the patient’s time and physical well-being—the Indian government is moving toward a more sustainable public health model. The ICMR-NIRT study provides the “evidence-based green light” that policymakers have been waiting for to finalize the programmatic rollout of these regimens nationwide.
Study Summary at a Glance
| Feature | Standard Long Regimen | BPaL/BPaLM Regimen |
| Duration | 18–20 Months | 6 Months |
| Administration | Oral + (Previously) Injectable | 100% Oral |
| Cost-Effectiveness | Baseline | Cost-Saving (BPaL) |
| Patient Adherence | Moderate to Low | High |
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
-
Study Citation: ICMR–National Institute for Research in Tuberculosis (ICMR-NIRT). “Cost-effectiveness of BPaL and BPaLM regimens for MDR/RR-TB in India.” Indian Journal of Medical Research (2026). DOI: [Accessed via ijmr.org.in/cost-effectiveness.pdf]