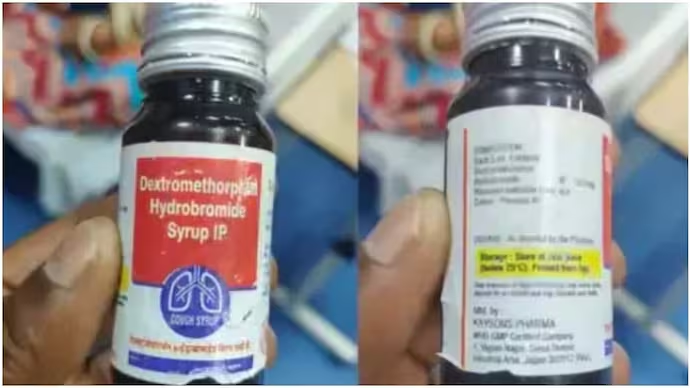
The National Human Rights Commission (NHRC) has launched a probe into the deaths of at least 12 children across Madhya Pradesh and Rajasthan, allegedly linked to the consumption of spurious cough syrup, sparking nationwide concern over drug safety and regulatory oversight in India.
What Happened? Who is Involved?
In early October 2025, the NHRC issued urgent notices to the Principal Secretaries of Health Departments in Madhya Pradesh, Rajasthan, and Uttar Pradesh, following a complaint that flagged serious lapses in the drug regulatory system. The deaths were reportedly concentrated in Chhindwara and Vidisha districts of MP, as well as districts in Rajasthan, with several cases pointing to kidney failure and other complications after children consumed locally distributed cough syrups.
Key regulatory bodies, including the Drugs Controller General of India (DCGI), the Central Drugs Standard Control Organisation (CDSCO), and the Union Health Ministry, have been directed to launch comprehensive investigations into both the supply chain and quality assurance of the implicated syrups.
Key Findings and Developments
-
Toxic Ingredients Identified: Initial tests revealed that certain batches of Coldrif syrup, manufactured by Sresan Pharma in Tamil Nadu, contained toxic levels of diethylene glycol (DEG), a substance known to cause acute kidney injury and death in children—a finding echoed in several international incidents involving Indian-made syrups.
-
Wider Product Bans: The sale of Coldrif syrup and analogous products has been temporarily banned in MP, Rajasthan, Uttar Pradesh, Karnataka, and Tamil Nadu, with other states such as Gujarat launching their own probe into harmful ingredients present in cough syrups.
-
Regulatory Oversight: The NHRC’s intervention points to systemic lapses in drug safety monitoring, suggesting that failures occurred not only at the manufacturing stage but also in regulatory and distribution channels.
- Expert Commentary
Dr. Rajiv Gupta, a pediatrician at PGIMER, Chandigarh, emphasizes, “Diethylene glycol poisoning is rare but extremely dangerous, as its early symptoms mimic viral illnesses, making prompt recognition difficult in pediatric settings. Strengthening laboratory testing and recall protocols is essential to prevent future tragedies”.
The Indian Medical Association (IMA) has criticized the arrest of a prescribing physician, arguing, “Quality control must happen at the manufacturer and regulatory level, not be shifted entirely onto prescribing physicians. The onus is on manufacturers and authorities to ensure safe medicines reach patients”.
Context and Background
This incident is not isolated; India has previously faced international scrutiny after contaminated cough syrups exported to Gambia, Cameroon, and Uzbekistan caused fatal poisonings among young children. The WHO has called attention to the problem, urging stricter controls and better surveillance over pharmaceutical ingredients and production standards.
The Union Health Ministry recently confirmed risk-based inspections at 19 pharmaceutical manufacturing units across six states, following these deaths. Additionally, a directive was issued that cough and cold medications should not be prescribed to children under two, due to their heightened risk of severe adverse reactions.
Implications for Public Health
-
Prescription Caution: This crisis underscores the need for healthcare professionals to exercise caution in prescribing over-the-counter cough syrups, especially to children under two years old.
-
Consumer Awareness: Parents should only use medications prescribed by licensed practitioners and avoid purchasing syrups from unregulated sources, verifying batch numbers when possible.
-
Regulatory Reforms: Experts suggest the need for enhanced post-marketing surveillance, traceability of ingredients, and mandatory rapid reporting of adverse drug reactions.
Counterarguments and Study Limitations
While toxic diethylene glycol was found in some batches, initial testing by the Union Health Ministry failed to detect contaminants in several others, indicating the possibility of batch-specific contamination. Some experts caution against blanket bans, as not all cough syrups pose the same risk, and abrupt removal could limit access to essential medications for those who genuinely need them.
Practical Takeaways
-
For Parents: Always consult a qualified healthcare professional before giving cough syrup to children.
-
For Providers: Dispense only medications with verified safety profiles; prioritize manufacturer information from reputable sources.
-
For Policymakers: Implement routine inspection schedules and strengthen penalties for drug adulteration to deter future lapses.
Medical Disclaimer
“Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.”
References
- https://www.newsonair.gov.in/nhrc-issues-notices-to-mp-rajasthan-and-up-over-child-deaths-linked-to-contaminated-cough-syrup/
- https://timesofindia.indiatimes.com/city/bhopal/nhrc-issues-notices-to-mp-raj-up-govts/articleshow/124347859.cms