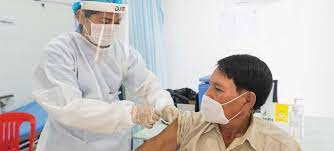
AJMER, RAJASTHAN — In a transformative move for women’s health in India, Prime Minister Narendra Modi officially launched the National Human Papillomavirus (HPV) Vaccination Campaign on February 28, 2026. Speaking from Ajmer, Rajasthan, the Prime Minister framed the initiative as a critical intersection of healthcare, education, and female empowerment.
The campaign specifically targets 14-year-old girls across the country, aiming to protect an estimated annual cohort of 1.2 crore (12 million) adolescents from cervical cancer—a disease that remains one of the leading causes of cancer-related mortality among Indian women. Rajasthan has been positioned as the “launchpad” for this mission, with over 188 vaccination sessions already conducted and approximately 1,000 institutions prepared to facilitate the rollout.
The Burden: Why This Campaign is a Public Health Priority
Cervical cancer is the second most common cancer among women in India. According to the World Health Organization’s (WHO) GLOBOCAN 2022 estimates, India recorded over 1.2 lakh new cases and nearly 80,000 deaths annually.
Scientific consensus identifies persistent infection with high-risk HPV types—specifically types 16 and 18—as the primary cause of the vast majority of cervical cancers.
“From a public health standpoint, HPV vaccination is one of the most powerful cancer-prevention tools we have for women,” says a senior gynaecologic oncologist at a prominent North Indian government cancer center. “If we can reach girls before they are exposed to the virus, we can dramatically cut future cervical cancer cases over the next two to three decades.”
Operational Roadmap: How the Vaccine Will Be Delivered
The national program has adopted a single-dose schedule using Gardasil-4, a quadrivalent vaccine that protects against HPV types 6, 11, 16, and 18. This approach follows 2022 WHO recommendations suggesting that a single dose provides robust, long-lasting protection comparable to multi-dose regimens, greatly simplifying the logistics of a nationwide scale-up.
Key Features of the Rollout:
-
The 90-Day “Campaign Mode”: The vaccine will initially be available daily at designated government centers for three months, after which it will be integrated into routine immunization schedules (typically twice weekly).
-
Access Points: Vaccination is free of charge at Ayushman Arogya Mandirs (PHCs), Community Health Centres, District Hospitals, and Government Medical Colleges.
-
Digital Integration: The U-WIN platform—a digital backbone modeled after the successful Co-WIN system—will manage registrations, consent, and vaccination records.
-
Cold Chain Integrity: Vaccines are stored between 2°C and 8°C, utilizing the existing robust infrastructure of India’s Universal Immunization Programme (UIP).
Consent and Safety: Navigating Adolescent Health
A significant departure from other routine childhood vaccines in India is the requirement for “opt-in” consent. Unlike the “opt-out” model where consent is generally assumed, parents or guardians must actively provide permission via the U-WIN app or on paper.
“The opt-in model respects family autonomy and cultural context,” notes a public health ethicist at a leading Indian medical college. “However, it places the onus on the government to ensure communication is transparent to avoid lower coverage due to simple lack of awareness.”
Regarding safety, Gardasil-4 has been administered to millions globally. Reported side effects are typically mild, including:
-
Pain or redness at the injection site
-
Low-grade fever
-
Temporary fatigue
All sessions are supervised by trained medical officers to manage any rare Adverse Events Following Immunization (AEFI).
Evidence of Impact: A Look at Global Success
Data from the 160 countries that have already introduced HPV vaccines show a 93% to 100% efficacy rate in preventing cervical cancer when administered before viral exposure. In nations with high coverage, there have been dramatic declines in both HPV infections and precancerous cervical lesions.
However, experts caution that the vaccine is a preventive measure, not a cure. “The science is clear that vaccinating before sexual debut yields the strongest protection,” says an infectious disease specialist in Delhi. “But we must emphasize that vaccination does not replace the need for cervical cancer screening (Pap smears or HPV tests) later in life, as the vaccine does not cover every possible cancer-causing HPV strain.”
Information for Families and Caregivers
For parents navigating this new health offering, the Ministry of Health has outlined clear eligibility and procedural guidelines:
| Feature | Details |
| Eligibility | Girls who have completed 14 years but not yet 15. |
| Cost | Free at all government facilities. |
| Documentation | Registration via U-WIN; left index finger may be marked with ink to prevent duplication. |
| Exclusions | Girls who have already received a full course of other HPV vaccines (e.g., Cervavac or Gardasil-9). |
Health authorities advise parents to consult their healthcare provider if their child has a history of severe allergies or is currently suffering from an acute illness.
Challenges and Future Outlook
Despite the scientific backing, the campaign faces the perennial challenge of vaccine hesitancy. Misinformation regarding fertility and safety often surfaces with adolescent-targeted vaccines. Public health officials are working to frame the shot strictly as cancer prevention rather than a commentary on behavior.
The long-term success of the drive will depend on community engagement, particularly in underserved regions. Rajasthan’s experience in using outreach strategies for tribal districts—such as those used during COVID-19—will be vital in reaching the “last mile.”
“When we protect girls today, we protect mothers and families tomorrow,” Prime Minister Modi stated during the launch. By integrating this vaccine into the national framework, India moves one step closer to the global goal of eliminating cervical cancer as a public health threat.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References