Chennai, Nov. 7 — Researchers at the Indian Institute of Technology Madras (IIT-M) have developed a patented, minimally invasive continuous glucose monitoring (CGM) patch that shows real-time glucose levels directly on a wearable display, combining reusable electronics with a disposable microneedle sensor to reduce costs and improve ease of use, according to the institute’s press release and corroborating reports from Indian media outlets. The device, designed by the Electronic Materials and Thin Films Lab led by Prof. Parasuraman Swaminathan, prioritizes low power consumption to extend battery life and is moving toward clinical validation and commercialization via a new startup while IIT-M engages with medical device manufacturers for licensing.
What’s new
-
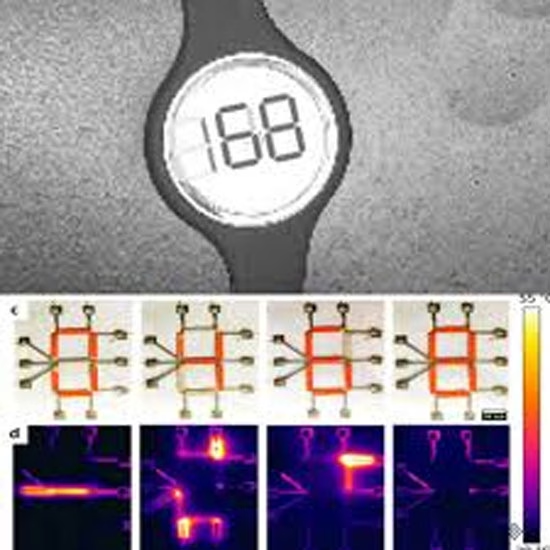
The IIT-M device integrates a reusable control unit and a novel low-power display with a disposable microneedle patch so glucose readings appear on the wearable itself without needing a phone or handheld reader, addressing cost and usability barriers cited for current CGMs.
-
Reports describe a patented electro-thermochromic display that consumes power mainly when updating readings, a design choice intended to lengthen battery life and reduce charging frequency compared with conventional screens used in many medical wearables.
-
The team has secured multiple Indian patents and filed an international PCT application, with functional prototypes tested and a startup at IIT-M Research Park established to refine the device and conduct clinical trials ahead of market entry, alongside parallel licensing talks with industry.
Why it matters
-
India has an estimated 10.1 crore (101 million) people living with diabetes, underscoring the need for scalable and affordable glucose monitoring options that support tight glycemic control and reduce complications, as referenced by IIT-M from the ICMR–INDIAB study published in 2023 in The Lancet Diabetes & Endocrinology.
-
While finger-prick self-monitoring of blood glucose (SMBG) remains common, it can be painful and cumbersome; CGM systems provide continuous, real-time trends but are often expensive and may require phones or proprietary readers—barriers that this patch’s direct-on-device display seeks to overcome.
-
A domestically developed CGM platform could also reduce dependence on imports, aid local manufacturing, and enhance self-reliance in medical technology, according to IIT-M’s public communications.
Expert viewpoints
-
Prof. Parasuraman Swaminathan emphasized translation to impact, noting that sparing patients multiple daily finger pricks and helping them “stay in range more consistently” could reduce long-term complications, if validated in clinical settings.
-
L. Balamurugan, an IIT-M MS (Entrepreneurship) scholar on the project, said a painless, discreet, affordable device may encourage more regular monitoring and timely action, potentially reducing emergencies and healthcare burdens at family and system levels.
-
Independent perspective: Clinicians typically assess CGMs on accuracy (often reported as mean absolute relative difference, MARD), reliability of interstitial-to-blood glucose correlation, calibration needs, wear time, and alarm features; these parameters, along with human factors and cost-effectiveness, will be crucial in forthcoming clinical trials for the IIT-M device, even though specific accuracy metrics have not yet been disclosed publicly.
How it compares
-
Current CGMs in the market generally use a subcutaneous filament sensor with wireless transmission to a smartphone or dedicated receiver; IIT-M’s approach embeds a low-power display so readings are visible on the patch itself, potentially simplifying workflows for users who prefer not to rely on phones or separate readers.
-
By modularizing the system—reusable electronics plus a disposable microneedle sensor—total cost of ownership could be lowered over time, provided the disposable component remains inexpensive and the reusable module is durable, which commercialization efforts will need to validate.
-
Low-power electro-thermochromic display technology aims to lengthen battery life, a frequent pain point in wearables, though real-world performance, readability in diverse lighting, and update cadence will need evaluation in trials.
Public health context
-
Improved access to CGM can help people with type 1 diabetes and insulin-treated type 2 diabetes recognize trends, limit hypoglycemia, and adjust therapy more safely, particularly when supported by education and clinical oversight—benefits that depend on device accuracy, reliability, and user adherence.
-
If the IIT-M device proves accurate and affordable at scale, it could support broader adoption in resource-constrained settings, complementing national efforts to manage the growing diabetes burden and potentially reducing hospitalizations tied to glucose excursions.
-
Domestic innovation and manufacturing may also mitigate supply chain constraints and lower costs for Indian users, aligning with Make in India goals and enhancing device availability for public and private health programs.
Limitations and open questions
-
Accuracy data, including MARD, calibration frequency, and lag characteristics between interstitial and blood glucose, have not yet been published in peer-reviewed venues; clinical trial outcomes will be essential for clinical acceptance.
-
Regulatory pathway and timelines (e.g., CDSCO approvals in India) remain to be clarified publicly; successful commercialization will hinge on meeting safety, performance, and quality system standards.
-
Durability of the reusable unit, cost of the disposable microneedle patch, wear duration, skin tolerance, and data connectivity options (if any) have not been fully detailed, and will influence real-world uptake across diverse user groups.
Practical takeaways for readers
-
For people monitoring glucose, a patch that shows readings directly on-device could reduce dependence on smartphones and make quick checks more convenient, especially for those seeking less intrusive solutions—pending clinical validation and regulatory approvals.
-
Even with promising technology, treatment changes should not be made based on a new device until validated by clinicians; users should continue current monitoring and therapy plans and discuss new options with healthcare providers as clinical trial data emerge.
-
Watch for forthcoming trial results, pricing, and availability announcements from the IIT-M startup or licensing partners, which will inform whether this device fits personal needs relative to existing CGM brands and SMBG routines.
What’s next
-
IIT-M’s startup will refine the device and conduct clinical trials, while the institute explores technology transfer and licensing with medical device manufacturers to accelerate market entry and scaling.
-
Public updates from IIT-M indicate momentum toward clinical validation and commercialization, with multiple patents already filed to protect the core display and system design innovations.
-
Media and institutional coverage suggest interest from digital health stakeholders, but peer-reviewed performance data will be the key milestone for healthcare professionals and payers assessing clinical adoption.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
- https://www.iitm.ac.in/happenings/press-releases-and-coverages/iit-madras-researchers-develop-affordable-minimally
- https://www.newindianexpress.com/states/tamil-nadu/2025/Nov/07/iit-m-develops-patented-affordable-painless-glucose-monitoring-device