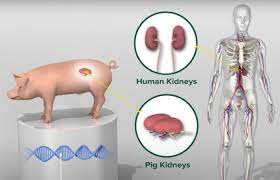
The first clinical trial for pig kidney transplants in humans has recently begun in the United States, marking a major milestone in xenotransplantation aimed at addressing the critical shortage of human donor organs. This pioneering trial specifically involves genetically modified pig kidneys designed to overcome biological rejection barriers, with the goal of providing a new treatment alternative for patients with end-stage renal disease who have limited access to human donor kidneys. The initial transplant surgery for the trial was reported at NYU Langone Health, led by Dr. Robert Montgomery, an early innovator in this field whose team has previously performed compassionate-use xenotransplants.
Genetically engineered pigs are central to this advance. The pig donors undergo precise gene editing—removal of four porcine genes that provoke rejection and insertion of six human genes to make the organs more compatible with the human immune system. This approach aims to prevent hyperacute rejection that historically doomed pig-to-human transplant attempts. The trial monitors recipient survival, kidney graft survival, kidney function via glomerular filtration rate (GFR), quality of life changes, and safety endpoints such as adverse events and infection risks over a 24-week post-transplant period.
Experts emphasize the significance of these gene modifications. Prof. Toby Coates of the University of Adelaide highlights that the trial kidney functioned immediately and showed no rejection signs for seven days in a brain-dead human donor, using standard immunosuppressive drugs. The trial’s lead at United Therapeutics points to the potential that pig kidney xenotransplantation holds in reducing lifelong dialysis dependency and expanding transplant access for patients unlikely to receive a human kidney.
Background clinical research also underscores the promise and challenges of pig kidney xenotransplants. Compassionate-use cases, such as a gene-edited pig kidney that strongly functioned for nearly nine months in a living human recipient, demonstrate feasibility but reveal durability questions. The first half of 2025 brought rapid progress—from laboratory advances to clinical translation—supported by regulatory frameworks like the FDA’s Expanded Access pathway, which facilitate trial designs prioritizing patient safety and informed consent.
However, experts caution that xenotransplantation is still in an early phase. Key questions remain about immune modulation for long-term acceptance, risks of zoonotic infections, and scaling production of gene-edited donor pigs. Ethical and regulatory challenges accompany these scientific advances, with international guidelines evolving to ensure transparency and equitable access as the field expands.
For the public and patients, the implications are profound. Kidney xenotransplantation could revolutionize treatment for over 100,000 patients on waitlists in the US alone, potentially reducing the burden of dialysis and shortening transplant waiting times. Yet, the community must balance hope with cautious optimism, recognizing that widespread clinical application requires more data on safety, efficacy, and long-term outcomes.
Key Developments in Pig Kidney Xenotransplantation
-
The first FDA-sanctioned clinical trial of genetically modified pig kidneys in humans began in late 2025 at NYU Langone Health.
-
Pigs are genetically modified to remove rejection-causing genes and add human genes to improve compatibility.
-
The trial tracks survival, kidney function, quality of life, and adverse effects over six months.
-
Compassionate-use cases showed pig kidneys working well for up to nine months in humans.
-
Standard immunosuppressive drugs are used to manage rejection in these transplants.
Expert Commentary
Prof. Toby Coates (University of Adelaide, Director of Transplantation):
“This trial provides proof of principle that genetically modified pig kidneys can replace human kidney function at least temporarily and without hyperacute rejection. The combination of gene editing to ‘humanize’ the organs and contemporary immunosuppressive therapy is a major breakthrough for kidney xenotransplantation.”
Dr. Leigh Peterson, Executive VP at United Therapeutics:
“Our goal is to expand access to transplantable organs beyond human donors, offering new hope to patients with end-stage renal disease. Patient safety and scientific rigor remain paramount as we advance this promising new therapy.”
Context and Background
Kidney failure is a global health challenge with over 800 million people affected worldwide. Organ shortages lead many patients to endure prolonged dialysis, which impacts quality of life and survival. Xenotransplantation—the transplantation of animal organs into humans—has long been considered a potential solution, but initial attempts failed due to rapid organ rejection. Advances in gene-editing technologies like CRISPR have re-energized this field, allowing precise genetic modifications to pig donors to reduce immune rejection risks.
This trial builds on multiple previous experimental and compassionate-use cases, including pivotal surgeries performed in brain-dead patients and living humans, demonstrating the capacity of pig kidneys to function in the human body.
Public Health Implications
If successful, pig kidney transplants could significantly reduce transplant waiting lists, improve survival rates, and lower healthcare costs associated with dialysis. This would represent a major public health breakthrough for kidney disease worldwide.
However, careful monitoring for potential zoonotic infections and immunological complications is essential, and health authorities will need to establish robust screening and follow-up protocols.
Limitations and Challenges
-
Early clinical trials are limited in size and duration; long-term graft survival and safety remain uncertain.
-
Ethical considerations, including informed consent and animal welfare, require ongoing scrutiny.
-
Scaling production and regulatory approval of genetically engineered donor pigs present logistical hurdles.
-
Public acceptance and education will be critical to successful implementation.
Medical Disclaimer
This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.