MUMBAI — Health experts and international regulatory bodies are calling for a critical review of rabies vaccinations administered over the past 14 months following the discovery of counterfeit vaccines circulating in the Indian market. The advisory, which targets individuals treated for animal bites or scratches since November 2023, emphasizes the life-saving necessity of ensuring vaccine authenticity in the face of a disease that remains 100% fatal once symptoms manifest.
The alert gained significant traction following a December 19 statement from Australia’s Technical Advisory Group on Immunisation (ATAGI) and subsequent warnings from prominent Indian medical professionals. Authorities are urging patients to verify their medical records, as the suspected counterfeit products may lack the necessary biological components to provide immunity against the rabies virus.
Understanding the Risk: Why Authenticity Matters
Rabies is a viral zoonotic disease that attacks the central nervous system. While it is entirely preventable through timely Post-Exposure Prophylaxis (PEP), the margin for error is non-existent.
“There is no need for widespread panic, but we must treat this with the seriousness it deserves because rabies is fatal once symptoms appear,” says Dr. Aniket Mule, a consultant in internal medicine at KIMS Hospitals, Thane. “A counterfeit vaccine isn’t just a financial fraud; it is a failure of a life-saving intervention. Prevention is our only tool.”
The counterfeit alert primarily concerns a specific brand of vaccine where the formulation, labeling, and manufacturing standards did not match the registered product. While the manufacturer acknowledged an “isolated case of non-original packaging” in January 2025 and has since cooperated with law enforcement to withdraw the affected lots, an unknown number of doses may have been administered before the recall.
Who Should Be Concerned?
Health authorities have narrowed the window of concern to vaccinations administered between November 2023 and January 2025. You should consult a healthcare provider if:
-
You received a rabies vaccine following an animal exposure during this timeframe.
-
The brand or batch number of the vaccine you received is unknown.
-
Your medical records match the specific batches flagged by regulatory authorities.
The Medical Response: To Revaccinate or Not?
The decision to revaccinate is not universal and requires a nuanced clinical assessment. Dr. Sivaranjani Santosh, a Hyderabad-based pediatrician who was among the first to flag the issue publicly, notes that individuals who can verify they received other approved brands do not need to worry.
“If your records show you received vaccines such as Vaxirab-N, Rabivax-S, Lyssavac, Vaxirab, or Indirab, you are considered protected and do not require repeat doses,” Dr. Santosh explains.
For those unsure of their vaccine’s origin, the clinical protocol involves several factors:
-
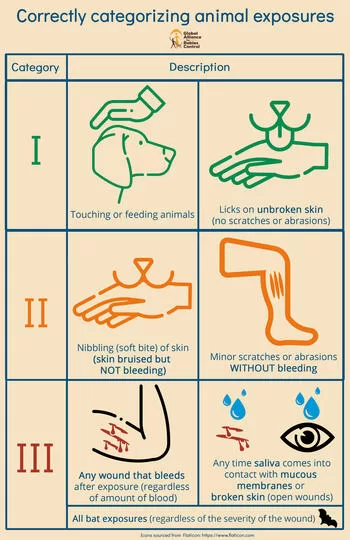
Nature of Exposure: Was it a deep bite (Category III), a minor scratch (Category II), or a lick on broken skin?
-
Administration of RIG: Was Rabies Immunoglobulin (which provides immediate antibodies) administered alongside the vaccine?
-
Vaccination Schedule: Was the full multi-dose course completed?
For high-risk individuals, such as veterinary professionals and animal rescuers who received Pre-Exposure Prophylaxis (PrEP), a blood test to check rabies antibody levels can be a definitive guide. If antibody levels are at or above 0.5 IU/mL, revaccination is generally not required.
Statistical Context and Public Health Implications
India carries a significant portion of the global rabies burden. According to the World Health Organization (WHO), India accounts for approximately 36% of global rabies deaths. The presence of counterfeit vaccines in such a high-endemic region poses a severe threat to public health.
The incubation period for rabies—the time between the bite and the onset of symptoms—is typically two weeks to three months, though in rare cases, it can extend beyond a year. This variability is why the current look-back period extends to late 2023.
“Because the incubation period can be lengthy, someone who received an ineffective vaccine months ago could still technically be at risk if the virus is traveling slowly through the nervous system,” explains Dr. Mule.
Counterarguments and Manufacturer Stance
In a statement released in early 2025, the manufacturer of the flagged brand emphasized that they have a “strong global safety record” and that the issue was an isolated incident of packaging tampering rather than a systemic failure of their genuine product. They noted that they proactively informed regulators and filed a criminal complaint to protect patients.
Some public health experts also caution against “over-vaccination.” They argue that if a patient is well past the six-month mark after a minor exposure and shows no symptoms, the physiological stress and cost of a new five-dose series should be weighed carefully by a physician. However, given the stakes, the prevailing medical consensus leans toward caution.
Practical Steps for Patients
If you or a family member received a rabies shot within the last 14 months, follow these steps:
-
Audit Your Records: Locate your vaccination card or hospital discharge summary. Look for the brand name and batch/lot number.
-
Contact the Clinic: If you don’t have records, contact the facility where the shot was administered. They are required to maintain logs of administered biologicals.
-
Consult a Specialist: Do not self-diagnose your risk level. See an internal medicine specialist or an infectious disease expert.
-
Check for Symptoms: While early symptoms are flu-like (fever, headache), seek emergency care for neurological signs like confusion, localized tingling at the bite site, or hydrophobia (fear of water).
Moving Forward: Strengthening the Supply Chain
This incident highlights a critical vulnerability in the global medical supply chain. Experts suggest that patients always ask to see the vaccine vial and packaging before administration and, where possible, take a photo of the batch number for their personal records.
“This is a wake-up call for more rigorous tracking and tracing of life-saving vaccines,” says Dr. Santosh. “Until then, the best defense for a patient is documentation and professional consultation.”
Medical Disclaimer
This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References and Sources
https://www.freepressjournal.in/mumbai/doctors-urge-review-of-rabies-shots-after-counterfeit-vaccine-alert