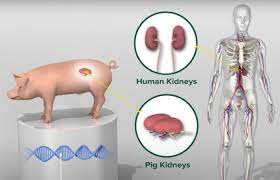
In a significant medical milestone, experimental transplants of genetically modified pig kidneys into human recipients in New Hampshire have renewed hope in addressing the critical shortage of donor organs. The recent surgery performed at Massachusetts General Hospital (Mass General) involved a 54-year-old man from Dover, New Hampshire, who is recovering well following his pioneering pig kidney transplant conducted on June 14, 2025. This achievement follows earlier successes, including another New Hampshire man who has lived for over seven months with a transplanted gene-edited pig kidney, setting a new survival record for such xenotransplants.
Advances and Key Developments
The experimental procedure involves xenotransplantation — the transplantation of organs from one species into another — utilizing kidneys sourced from genetically modified pigs. These pigs have undergone extensive genetic engineering, including the use of CRISPR technology to remove genes that produce substances triggering immediate rejection by the human immune system, such as the alpha-gal carbohydrate. Moreover, human genes have been inserted to increase compatibility, and viruses commonly present in pigs have been inactivated to reduce infection risks.
The pioneering biotech company eGenesis developed these gene-edited pigs specifically for organ transplantation. The U.S. Food and Drug Administration (FDA) has now authorized a large-scale clinical trial involving 30 patients aged 50 and above who are on dialysis and awaiting kidney transplants, marking a critical step toward more widespread clinical application.
Dr. Leonardo Riella, a kidney specialist at Mass General and co-leader of the upcoming trial, emphasized the urgency and potential impact of this work: “We currently have a bottleneck in sourcing sufficient human organs… over 100,000 individuals in the U.S. are waiting for transplants, the majority needing kidneys, with thousands dying while waiting”.Expert Commentary and Perspectives
Medical experts view recent successes as promising but caution that extensive evaluation remains necessary. Surgeon Dr. Tatsuo Kawai, director of Mass General’s Legorreta Center for Clinical Transplant Tolerance, lauded the recent pig kidney transplants, stating, “This second xenotransplant provides us with another excellent opportunity to learn how we can make genetically edited pig organs a viable, long-term solution for patients”.
Paul Conway, head of policy at the American Association of Kidney Patients, highlighted patient advocacy’s optimism toward these developments: “It’s a very hopeful time” as larger clinical trials commence.
However, ethical considerations remain integral to patient recruitment and trial design. Potential trial participants balance the risks of novel treatments against the mortality risk of remaining on the transplant waiting list. Concerns include the possibility of zoonotic infections transmitted from pigs to humans and the longevity and functionality of the pig organs long-term. Informing patients thoroughly before enrollment is a priority.
Background and Scientific Context
Kidney failure affects millions globally, with dialysis as a life-sustaining but quality-of-life-limiting therapy. The demand for kidney transplants far exceeds available donor organs. Waiting times can extend to several years, especially for patients with rare blood types. Reports indicate the 54-year-old transplant recipient had been on dialysis for over two years and faced long expected wait times for a matching human kidney.
Previous xenotransplant attempts in the U.S. have included pig kidney transplants at NYU Langone and pig heart transplants at the University of Maryland, conducted under compassionate use regulations, mainly for patients with no other treatment options. These cases provided vital early safety data but lacked the rigor of randomized clinical trials.
The gene editing approach used in these pigs encompasses about 69 genomic alterations aiming to maximize immune compatibility and reduce infection risk. Accompanying immunosuppressive drug protocols have been specifically designed to prevent rejection and have been provided by pharmaceutical collaborators.
Public Health Implications
If successful in clinical trials, genetically modified pig kidney transplants could revolutionize organ transplantation by substantially increasing organ availability. This could reduce waiting times, decrease dialysis dependency, and improve survival and quality of life for patients with end-stage renal disease.
Wider adoption of xenotransplantation could ease the burden on transplant systems worldwide and mitigate death rates among patients who expire waiting for human organs. Moreover, this approach may be eventually extended to other organs, addressing broader transplantation shortages.
Limitations and Future Directions
While recent cases show encouraging outcomes, the field faces challenges. Rejection remains a concern despite genetic modifications and immunosuppressive therapy, with some studies reporting episodes of T cell-mediated and antibody-mediated rejection detectable within days to weeks post-transplant. Reactivation of porcine viruses under immunosuppression has been documented, necessitating ongoing vigilance.
Long-term graft survival, comprehensive safety data, and evidence of functional equivalence to human donor kidneys await results from the upcoming clinical trials. Ethical issues regarding animal welfare, patient consent, and potential infectious risks are under careful review.
Practical Implications for Readers
For patients with kidney failure and their families, these advances represent a hopeful horizon where organ availability may improve significantly. However, pig kidney transplants remain experimental and are not yet a standard treatment option.
Patients should maintain communication with their nephrologists and transplant teams about emerging therapies and clinical trial opportunities. The breakthrough also underscores the importance of organ donation advocacy to bridge the current organ gap.
Medical Disclaimer
This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.