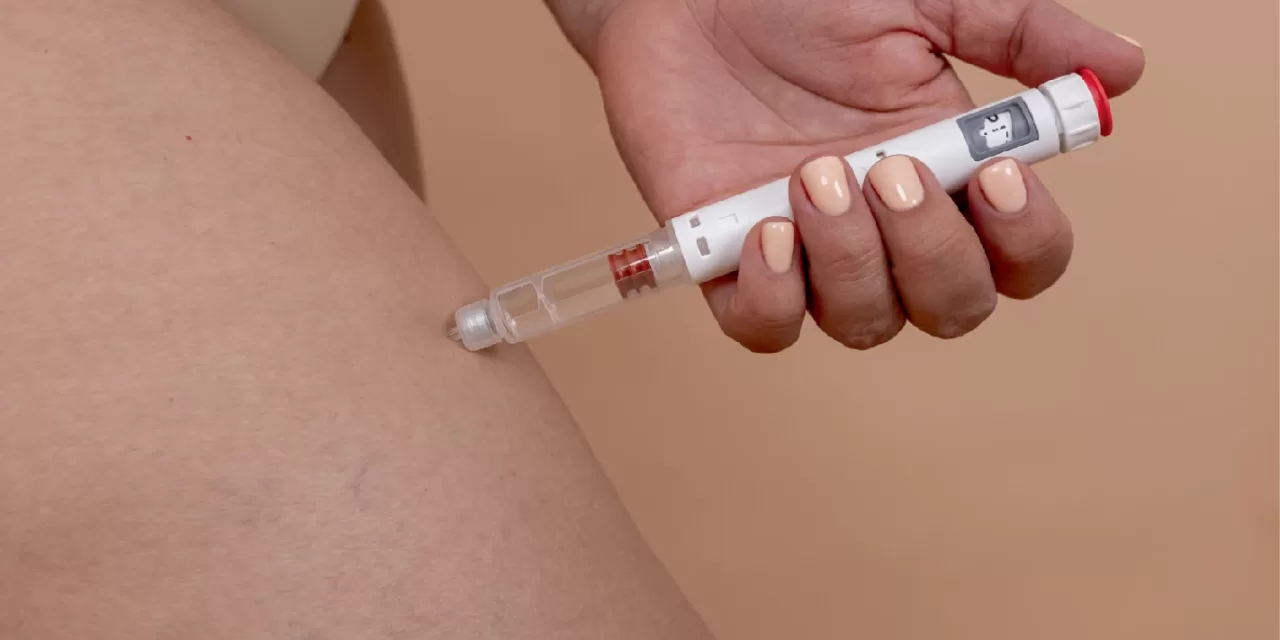
New York, Aug 16: In a significant health breakthrough, the U.S. Food and Drug Administration (FDA) has approved Novo Nordisk’s weight-loss drug Wegovy for the treatment of metabolic-associated steatohepatitis (MASH), a serious liver disease. This landmark decision grants Novo Nordisk a crucial head start over pharmaceutical rival Eli Lilly & Co. in the competitive U.S. market.
Wegovy, already a household name for weight management, is now cleared for use in adults afflicted with MASH who exhibit moderate to advanced liver scarring. The FDA estimates that about 14.9 million Americans—approximately 6% of the adult population—are impacted by this condition. With this approval, Wegovy’s use must be paired with a reduced-calorie diet and increased physical activity.
The move noticeably strengthens Novo Nordisk’s leadership in the rapidly growing GLP-1 drug market, positioning the company to fend off persistent rivalry from other pharmaceutical contenders and the looming threat of generic alternatives. The market reaction was swift: Novo Nordisk’s American depositary receipts surged 6%, while Madrigal Pharmaceuticals—another company pursuing treatments for MASH—saw its stock fall about 6.5%.
MASH is characterized by excessive fat accumulation in the liver, sparking inflammation and, eventually, potentially fatal complications like cirrhosis or liver cancer. Findings from clinical trials were promising: Wegovy improved liver fibrosis in 37% of treated patients, compared to 22% for those receiving a placebo. Additionally, nearly twice as many patients on Wegovy experienced a resolution of liver inflammation.
Martin Holst Lange, Chief Scientific Officer at Novo Nordisk, highlighted the scale of the issue, noting that roughly one in three overweight or obese individuals are affected by MASH. “Wegovy not only stops disease progression but also reverses liver damage in many patients,” Lange observed, underscoring the drug’s transformative potential.
This FDA approval could set the stage for broader utilization of GLP-1 therapies, extending beyond weight loss and diabetes to address complex liver diseases that have, until recently, lacked effective treatment options.
Disclaimer: This news article is intended for informational purposes only and should not be construed as medical advice. Please consult a qualified healthcare professional for personalized guidance regarding any health conditions or treatments.
Reference: Information compiled from recent FDA and pharmaceutical industry announcements as of August 16, 2025.