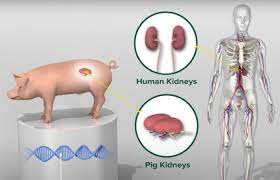
WASHINGTON D.C. – In a significant step for medical science, the U.S. Food and Drug Administration (FDA) has given the green light for a first-of-its-kind clinical trial to test the use of gene-edited pig livers as a temporary treatment for humans suffering from acute liver failure. The announcement was made Tuesday by eGenesis, the company producing the specialized pigs, in partnership with OrganOx.
The innovative approach will not involve transplanting the pig liver into the patient. Instead, the organ will be connected externally to the patient’s bloodstream. The goal is to have the pig liver filter the patient’s blood, performing the vital functions of a healthy liver, thereby giving the patient’s own damaged organ a crucial window to rest and potentially regenerate. The liver is unique in its ability to heal itself, and researchers hope this “bridge” therapy could provide the necessary time for recovery.
Acute liver failure is a life-threatening condition that leads to approximately 35,000 hospitalizations in the U.S. each year. Current treatment options are scarce, and mortality rates can be as high as 50%. Many patients are either not eligible for a liver transplant or cannot find a suitable donor organ in time.
This new trial aims to address this critical unmet need. It is expected to commence later this spring and will initially enroll up to 20 patients being treated in intensive care units who do not qualify for a conventional liver transplant.
During the procedure, the patient’s blood will be pumped through the external pig liver using a specialized device developed by UK-based OrganOx, which is already used for preserving donated human livers.
The pigs used in the trial are supplied by eGenesis, based in Massachusetts. The company employs genetic modification techniques to make the pigs’ organs more compatible with the human body, reducing the risk of rejection. Mike Curtis, CEO of eGenesis, mentioned that prior experiments involving four deceased human bodies indicated that the pig liver could successfully support some human liver functions for two to three days when connected externally.
This trial marks the latest advancement in the field of xenotransplantation – the use of animal organs for human treatment. It follows recent experimental transplants of gene-edited pig kidneys, also involving organs from eGenesis and another producer, United Therapeutics, showcasing growing momentum in efforts to use animal organs to save human lives.
Disclaimer: This news article is based on information provided by The Associated Press regarding an announcement by eGenesis and OrganOx concerning FDA clearance for a clinical trial. Further developments and trial results will determine the ultimate efficacy and safety of this potential treatment.
(Based on information reported by The Associated Press, © 2025)