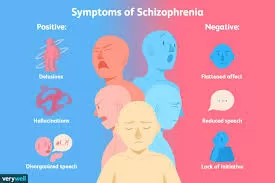
September 28, 2024 – Washington, D.C. — The U.S. Food and Drug Administration (FDA) has approved Cobenfy, a first-in-class antipsychotic developed by Bristol Myers Squibb, marking a significant milestone in the treatment of schizophrenia. Unlike traditional antipsychotics, Cobenfy targets cholinergic receptors in the brain, breaking away from the long-standing reliance on dopamine receptor antagonism, which has been the standard for over three decades.
Cobenfy’s approval follows promising clinical trials and introduces a combination of two key ingredients: xanomeline, a muscarinic cholinergic receptor agonist, and trospium chloride, a pan-muscarinic receptor antagonist. This dual approach aims to selectively target the M1 and M4 muscarinic receptors in the brain, offering a novel pathway to manage schizophrenia symptoms.
“A New Era in Schizophrenia Treatment”
“This drug represents the first new approach to treating schizophrenia in decades,” said Dr. Tiffany Farchione, MD, Director of the Division of Psychiatry in the FDA’s Office of Neuroscience. “It offers an alternative to treatments that rely on dopamine receptor blockade, bringing hope to patients who may not have responded well to traditional therapies.”
Cobenfy’s innovative mechanism could benefit patients who experience limited relief from current antipsychotic drugs. According to Ken Kramer, PhD, Vice President of Neuropsychiatry at Bristol Myers Squibb, “This drug avoids direct dopamine receptor blockade, distinguishing it from the current standards of care.”
Clinical Trial Success: Meeting Primary and Secondary Endpoints
The FDA’s decision was informed by data from the EMERGENT clinical program, which encompassed multiple efficacy and safety trials, including three placebo-controlled studies and two open-label studies on long-term safety.
In pivotal Phase 3 trials, EMERGENT-2 and EMERGENT-3, Cobenfy met its primary endpoint of significantly reducing schizophrenia symptoms, as measured by the Positive and Negative Syndrome Scale (PANSS). Patients treated with the drug showed an improvement in their total PANSS scores by 9.6 and 8.4 points, respectively, compared to those receiving a placebo. The trials also met all secondary endpoints, including reductions in the PANSS positive and negative subscales, along with improvements in the Clinical Global Impression-Severity (CGI-S) score.
Better Tolerability and Side-Effect Profile
In addition to its efficacy, Cobenfy offers a favorable side-effect profile compared to existing antipsychotics. While typical reactions included nausea, constipation, and dizziness, the drug notably did not cause weight gain or extrapyramidal symptoms, side effects often associated with older antipsychotics.
However, the FDA included warnings for potential adverse effects such as urinary retention, increased heart rate, and risks related to liver function. Patients with certain conditions, including kidney or liver impairment, urinary retention, or untreated narrow-angle glaucoma, are advised against using the drug.
Expert Reactions and Cautious Optimism
The approval has sparked widespread interest among experts in psychiatry. Dr. Sameer Jauhar, a consultant psychiatrist at King’s College, London, described the development as “possibly one of the most exciting advancements in schizophrenia treatment” due to its novel mechanism that bypasses the dopamine system.
Dr. Robert McCutcheon of the University of Oxford called the approval a “major advance,” emphasizing the need for new treatments to address symptoms not effectively managed by current drugs. However, some experts, including Dr. Paul Keedwell, a consultant psychiatrist, urge caution. While welcoming the innovation, Keedwell pointed out that further studies in real-world clinical settings are necessary to evaluate its long-term safety and tolerability, particularly regarding gastrointestinal side effects.
Availability and Patient Support
Cobenfy is expected to hit the market in late October, with capsules available in three dosage forms: 50 mg/20 mg, 100 mg/20 mg, and 125 mg/30 mg. To support patients, Bristol Myers Squibb has launched the “Cobenfy Cares” program, providing resources for those prescribed the drug.
As Cobenfy enters the schizophrenia treatment landscape, its impact will be closely monitored, potentially opening new therapeutic doors for patients struggling with this complex and debilitating disorder.