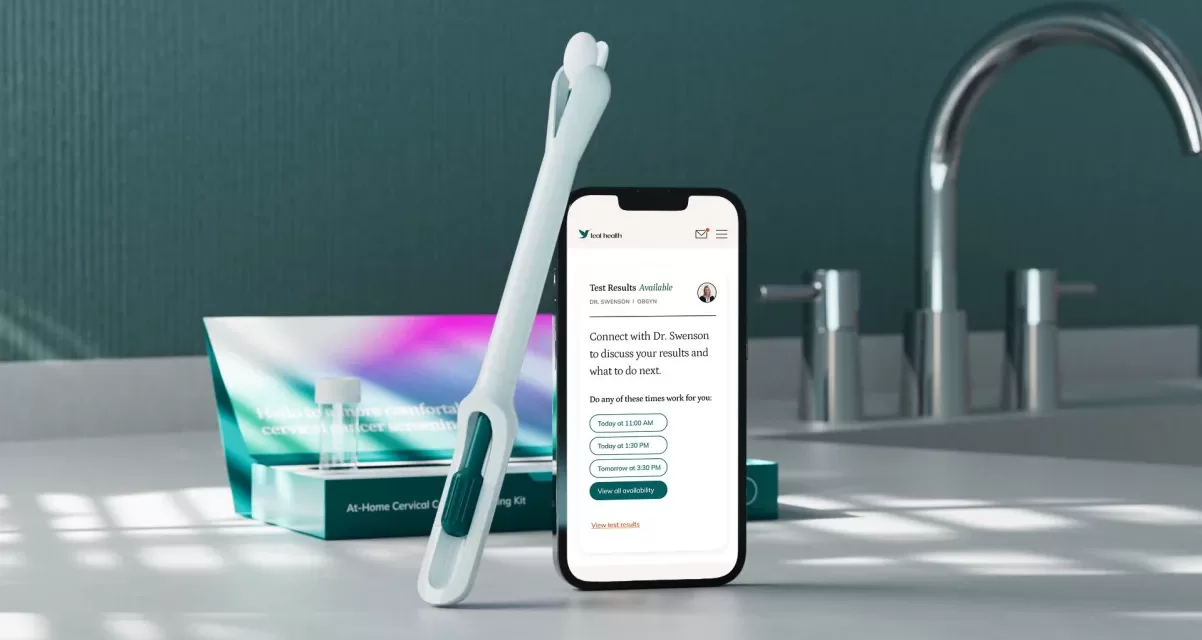
In a groundbreaking development for women’s health, the U.S. Food and Drug Administration (FDA) has approved the first at-home self-collection device for cervical cancer screening. The device, called the Teal Wand and developed by Teal Health, allows women to collect their own vaginal samples from the privacy of their homes, eliminating the need for in-person doctor visits and the discomfort often associated with traditional Pap smears.
How the Teal Wand Works
The Teal Wand will be available through Teal Health’s at-home self-collection kit, which requires a prescription. After requesting a kit on the Teal website and consulting with a healthcare provider, users can collect a sample at home and mail it to a laboratory for testing using the Roche Cobas HPV test. Results are reviewed by a clinician and then shared with the patient. If the test returns positive for high-risk human papillomavirus (HPV)-the primary cause of nearly all cervical cancers-a provider will follow up to discuss next steps.
Clinical trials involving over 600 women found that self-collected samples using the Teal Wand were as accurate for cervical cancer screening as those collected by healthcare professionals in clinical settings. “It’s the same test, same accuracy, but you can comfortably do it from home,” said Kara Egan, CEO and co-founder of Teal Health.
Why This Matters
Cervical cancer remains a significant health threat, with about 11,500 new cases and around 4,000 deaths annually in the United States, according to the Centers for Disease Control and Prevention. Regular screening is crucial for early detection and prevention, but many women delay or avoid Pap smears due to discomfort, inconvenience, or lack of access to healthcare providers. Experts believe the at-home option could help close these gaps, especially for those who have trouble scheduling appointments or live far from clinics.
Dr. William Dahut, chief scientific officer at the American Cancer Society, praised the FDA’s decision, stating, “Most cervical cancers are found in people who have never had a cervical cancer screening test or who have not had one recently. That’s why today’s FDA announcement approving the first at-home test… will make a huge impact”.
What’s Next?
Teal Health plans to launch the at-home kits in June, initially in California, with plans for nationwide expansion. The company is working with insurance providers to ensure coverage and will announce pricing for uninsured individuals soon. A waitlist is already available on the company’s website.
Screening Recommendations and HPV Facts
-
The U.S. Preventive Services Task Force recommends cervical cancer screening for women ages 21 to 65, with intervals varying based on age and test type.
-
HPV is a group of more than 150 viruses, and about 80% of people will contract an HPV infection in their lifetime. While most infections clear on their own, persistent high-risk strains can lead to cancer.
-
Early-stage cervical cancer often has no symptoms, making regular screening vital for early detection and effective treatment.
Disclaimer:
This article summarizes recent FDA approval and expert commentary regarding the Teal Wand at-home cervical cancer screening device. It is not a substitute for professional medical advice, diagnosis, or treatment. Individuals should consult their healthcare provider for guidance on cervical cancer screening and personal health decisions.
Citations: