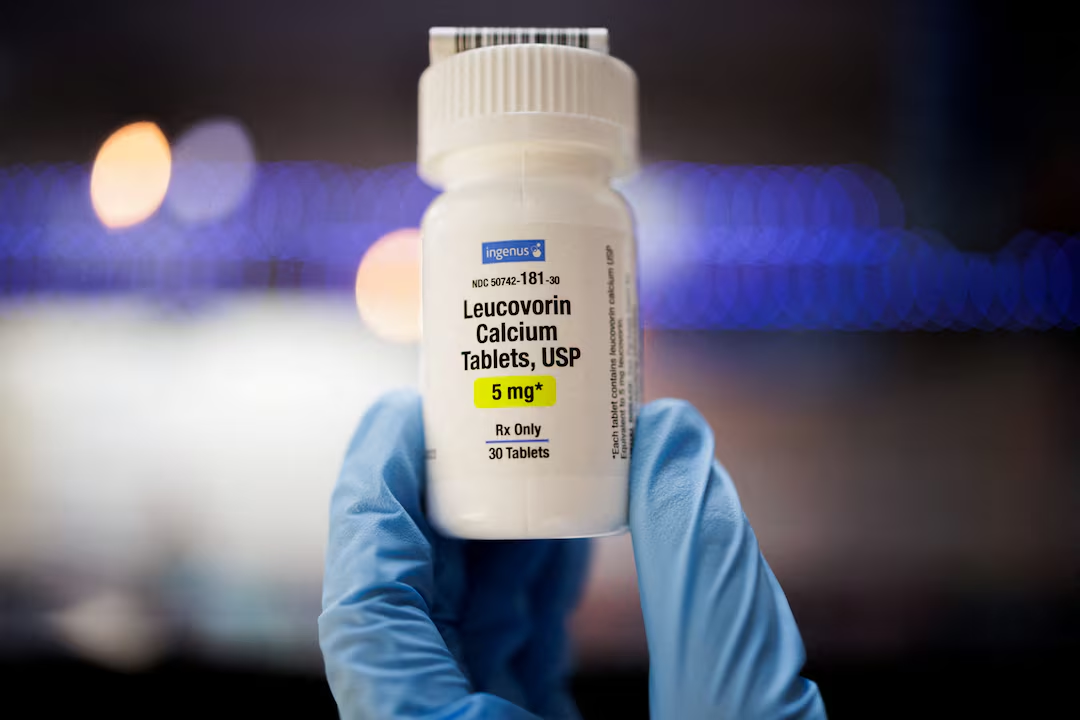
In a significant development dated September 22, 2025, the U.S. Food and Drug Administration (FDA) officially approved leucovorin calcium, a derivative of folic acid, as a treatment for cerebral folate deficiency (CFD)—a neurological condition linked to developmental delays and autism-like symptoms. This decision, endorsed publicly at a White House event shortly thereafter, has stirred intense interest among parents of children with autism, alongside cautious responses from medical experts and pediatricians.
Leucovorin’s link to autism revolves around its potential to address CFD, a condition impairing folate transport into the brain that has been observed in some individuals displaying neuropsychiatric symptoms associated with autism spectrum disorder (ASD). The FDA based its approval on patient-level data from over 40 individuals spanning adults and children, noting improvements in symptoms related to cerebral folate deficiency.
However, while the FDA’s move signals hope for families seeking new therapeutic options, the broader autism community, medical researchers, and professional organizations emphasize that current evidence for leucovorin’s effectiveness in treating autism at large remains limited. The American Academy of Pediatrics (AAP), for example, released guidance advising against the routine use of leucovorin in children with autism due to scant evidence on the benefits and potential risks, advocating instead for more rigorous studies to clarify safety, dosage, and patient selection.
Medical Experts’ Views and the Surge in Demand
The endorsement by then-President Donald Trump and FDA Commissioner Marty Makary substantially heightened public awareness and demand for leucovorin among parents of autistic children. Pediatricians report being inundated with requests for the drug, with some Facebook groups dedicated to sharing anecdotal success stories suddenly ballooning in membership to tens of thousands. While personal accounts of improved verbal skills and social awareness circulate widely, experts caution these testimonials do not replace scientifically robust clinical trials.
Dr. Kristin Sohl, an autism specialist involved in guideline development, noted the current uncertainties around who exactly might benefit from leucovorin, the optimal dosing strategy, and its safety profile. She advocates a cautious, shared decision-making process between pediatricians and families, with vigilant monitoring of treatment effects and side effects. Dr. David Mandell, an autism researcher, also remarked on the surge of parental interest alongside the need for careful evaluation of the data.
Context and Background on Cerebral Folate Deficiency and Autism
Cerebral folate deficiency is a rare metabolic condition that affects the transport of folate into the central nervous system, which is essential for brain function and development. Symptoms often overlap with features observed in autism such as challenges in social communication, sensory processing issues, and repetitive behaviors. However, CFD is distinct from autism spectrum disorder, which is a complex neurodevelopmental condition with multiple genetic and environmental factors.
Leucovorin has been used for decades as a folate replacement therapy in specific disorders and is known for its safety in approved indications. Its repurposing for CFD-related autism symptoms is an emerging area of clinical investigation. Unlike many newer experimental autism treatments, leucovorin benefits from an existing safety profile but lacks comprehensive clinical trial data in autism populations.
Public Health Implications and Considerations
The FDA’s approval of leucovorin for cerebral folate deficiency marks a strategic effort to repurpose an older drug for a rare but impactful neurological condition. For families affected by CFD-associated symptoms, this presents a potential new therapeutic avenue. Yet, it also raises broader questions about drug approval pathways for niche indications and the challenges of addressing the heterogeneous nature of autism.
Healthcare providers are urged to balance cautious optimism with scientific rigor. The American Academy of Pediatrics recommends minimizing potential harm through careful patient selection and close monitoring when prescribing leucovorin off-label. Furthermore, experts call for expanded clinical trials to elucidate which children might truly benefit and to establish standardized treatment protocols.
Counterarguments and Limitations
While enthusiasm runs high among parents, critics highlight that the evidence supporting leucovorin’s effect on core autism symptoms is currently insufficient. The data mainly derive from small case series and anecdotal reports, with no large-scale randomized controlled trials completed. This gap leaves unanswered questions about long-term safety, efficacy across the diverse autism spectrum, and interaction with other therapies.
Additionally, some caution against the politicization of medical decisions, pointing out that endorsements by political figures may advance treatments before the evidence base matures. Independent experts advocate maintaining stringent standards for evaluating emerging autism treatments to avoid false hope and potential side effects.
Practical Guidance for Families and Caregivers
For health-conscious readers and caregivers considering leucovorin:
-
Consult qualified healthcare professionals before starting any new treatment.
-
Understand that leucovorin is approved specifically for cerebral folate deficiency but not broadly for autism.
-
Seek care from providers experienced in autism and folate metabolism disorders who can guide informed decision-making.
-
Monitor for side effects and efficacy in a structured clinical setting.
-
Stay informed about ongoing research developments and clinical trials in this area.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References:
-
Reuters. FDA autism drug move sparks frenzy, but data lags behind. Michaelman et al., October 31, 2025. DOI unavailable.