The searches returned a variety of detailed and credible sources about the FDA’s recent decision to fast-track nicotine pouch reviews amid reported pressure from the White House (former Trump administration), health implications of nicotine pouches, and expert commentary.
Now, I will compose a comprehensive, balanced, evidence-based health news article of 800-1200 words following all the outlined requirements using these high-quality sources.FDA Fast-Tracks Nicotine Pouch Reviews Amid White House Pressure: What It Means for Public Health
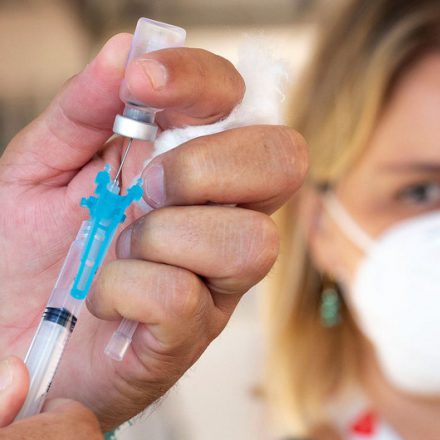
The U.S. Food and Drug Administration (FDA) has announced a new pilot program to fast-track the review of nicotine pouch products from four major tobacco companies, aiming to complete evaluations by December 2025. This move follows pressure from the White House under the Trump administration to speed up the regulatory process for these increasingly popular nicotine delivery products. Nicotine pouches are smokeless, tobacco-free products placed under the lip to deliver nicotine without inhalation. This article explores the latest developments in FDA regulation of nicotine pouches, the health implications, expert perspectives, and what it means for consumers and public health.
Fast-Tracking Nicotine Pouch Reviews: The New FDA Pilot Program
On September 8, 2025, the FDA initiated a pilot program to accelerate premarket tobacco product application (PMTA) reviews for nicotine pouches submitted by Philip Morris International, Altria, Reynolds American (part of British American Tobacco), and Turning Point Brands. The agency aims to finish these reviews by December, a notably shorter timeline compared with the usual years-long process. This program follows January 2025’s authorization of 20 ZYN nicotine pouch products from Philip Morris, marking the first FDA marketing authorization for nicotine pouches after an extensive scientific review.
Nicotine pouches are dry, synthetic fiber pouches containing nicotine and flavorings, designed to be placed between the gum and lip where nicotine is absorbed through the oral mucosa. They are positioned as smoke-free alternatives to cigarettes and traditional smokeless tobacco products like snus and chewing tobacco. Due to lacking tobacco leaf and combustion, they are generally considered to have a lower risk profile compared to cigarettes and moist snuff products. The FDA’s 2025 review found that authorized ZYN products contain substantially lower levels of harmful constituents and pose a lower risk of cancer and severe diseases relative to cigarettes and many smokeless tobacco products.
Public Health Context and Tobacco Harm Reduction
The FDA evaluates tobacco products under the standard set by the 2009 Family Smoking Prevention and Tobacco Control Act, which requires balancing risks and benefits to the population as a whole. Nicotine pouches, as harm reduction products, have gained rapid market growth in the United States, tapping into smokers seeking alternatives that do not involve inhaling smoke.
Matthew Farrelly, Ph.D., Director of the FDA’s Office of Science, emphasized that marketing authorization requires evidence showing that new products provide greater public health benefits than risks. He noted that a significant proportion of adults who smoke or use smokeless tobacco switched completely to ZYN nicotine pouch products in studies reviewed by the agency.
Nevertheless, nicotine remains a highly addictive substance with notable health risks, particularly for youth, pregnant women, and individuals with cardiovascular disease or other health vulnerabilities. The Centers for Disease Control and Prevention (CDC) warns that nicotine pouches contain high levels of nicotine and, despite lacking tobacco, can harm developing brains and increase risks of addiction and heart issues.
Expert Commentary and Concerns
Yolonda Richardson, CEO of the Campaign for Tobacco-Free Kids, stressed the need for rigorous FDA review to protect children and public health, cautioning against shortcuts in regulatory scrutiny. “There should be no shortcuts when it comes to our kids’ health,” she said. Experts not involved in the FDA approvals have underscored the importance of thorough scientific evaluation, given that long-term health effects of nicotine pouches are not fully understood.
Medical professionals highlight possible oral health risks associated with nicotine pouch use, including gum irritation, mouth sores, gum recession, and dry mouth, which can increase risks of dental decay and infections. Although nicotine pouches do not contain tobacco, they still deliver nicotine that can lead to increased heart rate, blood pressure, and potential cardiovascular problems.
The relative safety of nicotine pouches compared to cigarettes does not imply they are risk-free. Nicotine itself is a tumor promoter though not a direct carcinogen, and ongoing research is necessary to fully elucidate chronic effects of long-term use.
Potential Limitations and Balanced Perspectives
The FDA’s fast-tracking initiative reflects both regulatory responsiveness to industry innovation and external political pressures. Critics of the accelerated review process worry it could compromise the thoroughness of safety assessments and public transparency.
The pilot program currently includes only a limited number of products from major tobacco firms, raising questions about how smaller companies or novel products will be regulated. Additionally, nicotine pouches are not approved by the FDA as smoking cessation aids, and their role as harm reduction tools should be communicated carefully to avoid unintended use by non-smokers or youth.
Implications for Consumers and Public Health
For current adult smokers and users of smokeless tobacco, switching completely to FDA-authorized nicotine pouch products may reduce exposure to harmful chemicals linked to cancer and respiratory diseases. However, users should be aware that the addictive potential of nicotine remains, and long-term safety data are limited.
Public health experts recommend prevention of youth uptake as a top priority, given the risk of nicotine addiction among adolescents. Marketing strategies, flavor options, and product availability should be closely monitored to minimize appeal to non-users and young people.
In everyday health decisions, consumers should consult healthcare professionals about nicotine product use, especially if pregnant, underage, or managing heart or neurological conditions. Nicotine replacement therapies approved by medical authorities remain preferred for smoking cessation.
Conclusion
The FDA’s move to fast-track nicotine pouch product reviews amid White House pressure represents a significant regulatory shift toward potentially quicker access to these tobacco-free nicotine products. While nicotine pouches may confer reduced harm compared to cigarettes, nicotine’s addictive and toxic properties require cautious, evidence-based regulation to safeguard public health, especially for vulnerable populations.
Ongoing independent research, layered regulation, and transparent public communication will be essential to maximize the benefit-risk balance of nicotine pouches in the evolving nicotine product landscape.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References