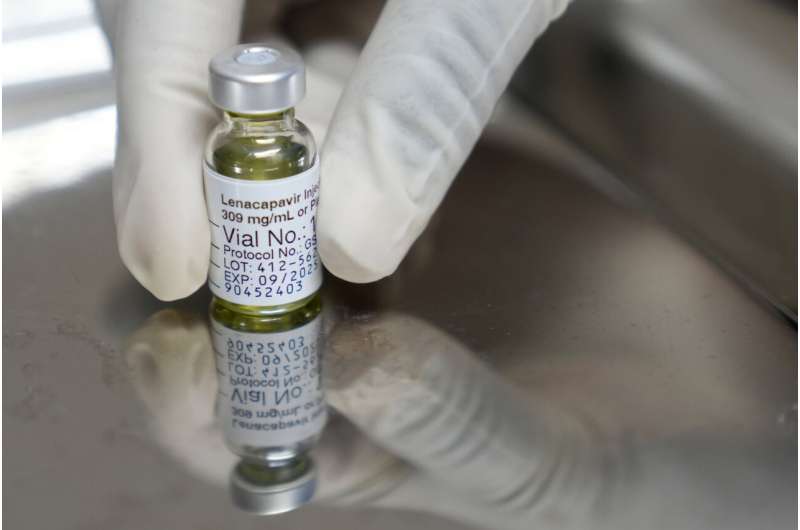
The European Commission (EC) has granted marketing authorization for Gilead Sciences’ novel twice-yearly injectable medication, Yeytuo® (lenacapavir), for pre-exposure prophylaxis (PrEP) against HIV infection. This decision on August 26, 2025, marks a significant advancement in HIV prevention across the European Union (EU), including its 27 member states as well as Norway, Iceland, and Liechtenstein. The approval comes shortly after U.S. regulatory clearance in June 2025, underscoring growing international confidence in this new prevention tool.
What Is Lenacapavir and Who Can Benefit?
Lenacapavir is a long-acting HIV-1 capsid inhibitor that is administered as a subcutaneous injection every six months. It is authorized for adults and adolescents at increased risk of sexually acquired HIV-1 infection who weigh at least 35 kilograms (approximately 77 pounds). Unlike daily oral PrEP medications, which often face adherence challenges, lenacapavir offers a convenient, biannual dosing schedule, which can transform prevention strategies for individuals who struggle with daily pill routines or face societal stigma.
Key Clinical Findings and Efficacy
The marketing authorization followed a rigorous, accelerated regulatory review by the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP), based on comprehensive clinical data from the PURPOSE program—a series of Phase 2 and 3 trials involving diverse global populations. Results from these studies demonstrated exceptional efficacy, with lenacapavir showing near 100% prevention of HIV infection. In one pivotal study, no infections occurred among those receiving the injection, and in another, effectiveness was measured at a 96% risk reduction compared to background incidence rates. It also showed superiority compared to the daily oral PrEP option Truvada® (emtricitabine/tenofovir disoproxil fumarate).
Dr. Dietmar Berger, Chief Medical Officer at Gilead Sciences, highlighted, “Yeytuo’s rapid authorization by the European Commission underscores the rigor of our clinical data and the transformative potential of Yeytuo to help address the urgent unmet need in HIV prevention across Europe. This milestone is a testament to 17 years of research by Gilead that delivered this breakthrough PrEP medication, supported by decades of leadership in HIV innovation”.
How Lenacapavir Works: A Novel Mechanism
Lenacapavir’s mechanism distinguishes it from other antiretrovirals by targeting multiple stages of the HIV lifecycle, rather than one single stage of viral replication. This multi-stage inhibition lowers the likelihood of resistance and enhances its preventative potency. Additionally, lenacapavir has no known cross-resistance with other currently approved HIV drug classes, which is promising for individuals at high risk or with existing resistance concerns.
Context and Implications for Public Health
HIV prevention efforts worldwide have plateaued in recent years, with the World Health Organization (WHO) estimating approximately 1.3 million new HIV infections projected for 2024. Vulnerable groups such as men who have sex with men, transgender individuals, people who inject drugs, incarcerated populations, adolescents, and other marginalized communities continue to experience disproportionate HIV transmission rates. The availability of lenacapavir injectables represents a critical new option to help reduce these rates by improving adherence and access challenges inherent to daily oral PrEP.
The WHO has endorsed lenacapavir as an HIV prevention tool, recognizing its potential to shift prevention paradigms particularly where stigma or logistical issues limit the use of daily pills. This approval advances global efforts to curb HIV transmission and meet public health targets.
Potential Limitations and Challenges
Despite the transformative potential of lenacapavir, challenges remain. Pricing and access are critical concerns, particularly in the EU and globally. In the US, the annual list price exceeds $28,000, which has led some insurers to delay coverage decisions. Similar pricing questions loom for European health systems, where national reimbursement negotiations must precede patient availability. Experts emphasize the importance of equitable pricing to avoid limiting access to this breakthrough medication.
Further, while clinical trials demonstrated excellent tolerability and safety profiles, as with any medication, ongoing pharmacovigilance and real-world experience are necessary to detect rare or long-term adverse effects.
Expert Perspectives
Professor Jane Anderson, an HIV specialist not affiliated with the studies, commented, “The approval of a six-month injectable PrEP option is a landmark moment in HIV prevention. It offers hope for greater adherence and reduced HIV spread, especially for those who find daily pills challenging. However, balancing cost and access will be crucial to realizing its full public health impact”.
Practical Implications for Readers
For individuals at higher risk of HIV, lenacapavir offers a new prevention choice with fewer doses and associated stigma compared to daily oral medication. Patients interested should consult healthcare providers specializing in sexual health or infectious diseases to explore whether this new injectable option fits their prevention goals and health profile. Public health programs may integrate this innovation to enhance community-wide HIV prevention strategies.
Medical Disclaimer
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.