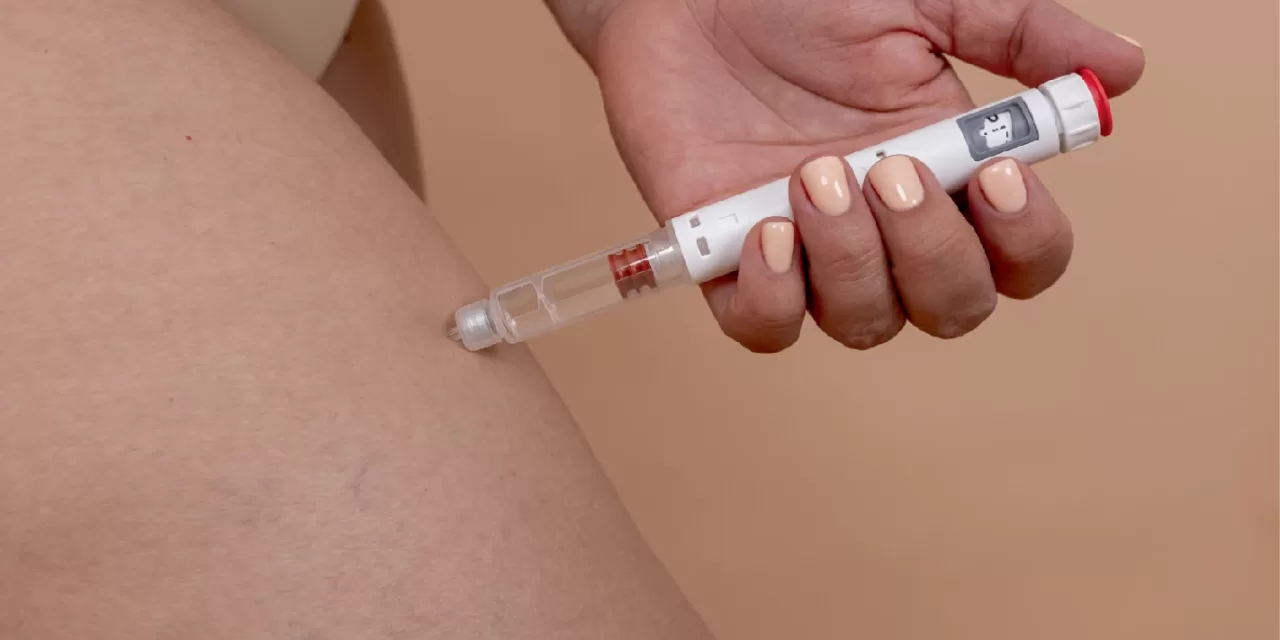
Eli Lilly’s latest experimental obesity drug has demonstrated significant weight loss in early-stage clinical trials, according to a report published on June 13, 2025. The new drug, currently in development, helped participants achieve over 11% weight loss in the initial phase of testing, raising hopes for a new treatment option in the ongoing battle against obesity.
The early trial results suggest the drug could become a major contender in the rapidly growing market for obesity treatments, which has seen increased competition and innovation in recent years. Eli Lilly, already known for its successful diabetes and weight loss medications, is aiming to build on its reputation by introducing another effective therapy for individuals struggling with obesity.
While these initial findings are encouraging, experts caution that further research is needed to confirm the drug’s safety and effectiveness over longer periods and in larger, more diverse populations. Regulatory approval will depend on the outcomes of additional clinical trials, which are expected to continue in the coming months.
“The results are promising, but we must wait for more comprehensive data before drawing conclusions about the drug’s long-term impact,” said a spokesperson familiar with the trial.
If successful, Eli Lilly’s new drug could offer another valuable tool for healthcare providers and patients seeking to manage obesity, a condition linked to numerous chronic health issues worldwide.
Disclaimer: This article is based on preliminary clinical trial data. The drug discussed is still under investigation and has not yet been approved for widespread use. Individuals should consult healthcare professionals for medical advice and not make any changes to their treatment based on early-stage research findings.