India’s drug safety authority has issued a new alert flagging important adverse reactions linked to two commonly used medicines, tranexamic acid and metoclopramide. This development highlights the ongoing need for diligent monitoring in clinical settings—especially as these drugs occupy a central role in various life-saving treatments and daily healthcare practice.
Background
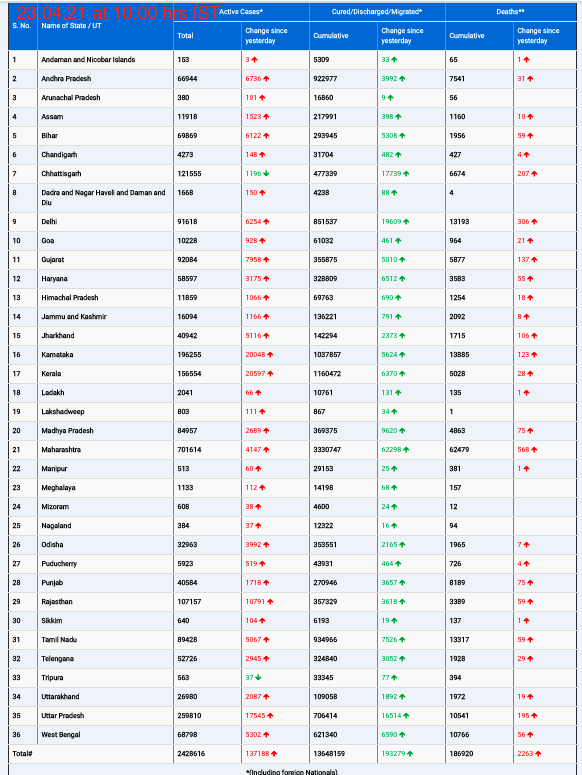
On September 6, 2025, the Indian Pharmacopoeia Commission (IPC), under the Ministry of Health and Family Welfare, released its latest drug safety alert, cautioning healthcare professionals and the public about newly identified adverse drug reactions (ADRs) for tranexamic acid and metoclopramide. The evidence comes from preliminary analyses in the Pharmacovigilance Programme of India (PvPI) database, which tracks drug-related safety signals nationwide.
Key Findings and Developments
-
Tranexamic Acid: Widely prescribed for treating and preventing excessive bleeding, tranexamic acid has now been associated with nasal congestion. This is not a trivial side effect—nasal congestion can impact breathing comfort and quality of life, especially in vulnerable populations needing this medication for surgeries, heavy menstrual bleeding, or bleeding disorders.
-
Metoclopramide: Used extensively as an antiemetic and prokinetic (for nausea, vomiting, and gastrointestinal issues), metoclopramide has shown a possible link with tachycardia—a medical condition where the resting heart rate exceeds 100 beats per minute and may put patients at risk for further cardiac complications.
Both findings come from India’s robust adverse event reporting system, which compiles patient, doctor, and pharmacist feedback into a centralized safety analysis. Although these ADRs are not universally experienced, the IPC recommends heightened vigilance every time these medicines are prescribed or administered.
Context and Background
Tranexamic Acid
-
Indications: Used to control bleeding in surgery, trauma, hemophilia, and gynecological disorders.
-
Mechanism: It works by inhibiting the breakdown of blood clots—a process called fibrinolysis—helping stabilize bleeding sites.
-
Market Use: Considered a regular feature in hospitals and clinics across India and globally, with its market projected to exceed $100 million in coming years.
Metoclopramide
-
Indications: Prescribed for nausea, vomiting (especially post-op and chemotherapy), and motility disorders like reflux and gastroparesis.
-
Mechanism: Acts via dopamine and serotonin receptor antagonism in the brain, promoting coordinated movement in the upper digestive tract.
-
Prevalence: Common in both inpatient and outpatient care, especially among those with gastrointestinal issues.
Expert Perspectives
Dr. Surbhi Sharma, MD (General Medicine), not involved in the PvPI analysis, explains: “Nasal congestion linked to tranexamic acid is relatively rare but important to note, especially in post-surgical or frail patients, where any additional airway compromise is concerning.” She adds that similar caution should be used with metoclopramide, which, despite being well-tolerated by most, can pose cardiac risks in sensitive individuals or the elderly.
Dr. Arjun Mehta, Consultant Pharmacologist, emphasizes: “Active pharmacovigilance—reporting every suspected ADR—enhances patient safety and builds a more resilient healthcare ecosystem. The IPC’s alert is a reminder for clinicians to never ignore even minor symptoms.”
Implications for Public Health
For consumers and practitioners, these alerts underscore several key implications:
-
Monitoring is Essential: Patients on tranexamic acid should report any persistent nasal congestion, while those prescribed metoclopramide should be vigilant for palpitations or rapid heartbeat.
-
Reporting Matters: ADRs, even seemingly minor ones, should be promptly reported using IPC’s online forms, helplines, or the dedicated ADR reporting mobile app.
-
Informed Consent: Conversations about medication risks should be routine, so patients understand both benefits and possible downsides before starting treatment.
Limitations and Counterarguments
-
Rarity of ADRs: Not every patient will experience these reactions; rates remain low relative to the number of people treated successfully. The observed ADRs emerged from risk signals—not conclusive causal proof.
-
Data Diversity: Causality can be challenging to establish. Nasal congestion and tachycardia can arise from myriad causes, and attributing them solely to the drugs may ignore underlying patient variability or other concurrent health conditions.
-
Global Relevance: Most global health agencies, including the FDA and WHO, still consider these medications safe when used according to guidelines—but stress the importance of country-specific pharmacovigilance protocols.
Practical Implications
-
For Health Professionals: Proactively monitor for and record any ADRs related to these drugs, ensuring patients are well-informed and receive follow-up if symptoms develop.
-
For Patients: Always disclose all symptoms during medication use, report any new or unusual experiences, and never discontinue medications without consulting qualified healthcare providers.
-
For Public Health Policymakers: Continued investment in pharmacovigilance training and public education campaigns will help capture ADRs earlier, prevent harm, and sustain public trust.
Balanced Reporting
While risk exists, both tranexamic acid and metoclopramide remain critical options in modern medicine. The emergence of documented ADRs does not invalidate their therapeutic value, but reinforces the need for closer observation, transparent conversations, and robust ADR reporting.
Medical Disclaimer
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
-
Indian Pharmacopoeia Commission (IPC), Drug Safety Alert: Adverse reactions linked to Tranexamic Acid and Metoclopramide. Medical Dialogues, September 6, 2025. https://medicaldialogues.in/news/industry/pharma/drug-safety-alert-ipc-flags-adverse-reactions-linked-to-tranexamic-acid-and-metoclopramide-154773medicaldialogues