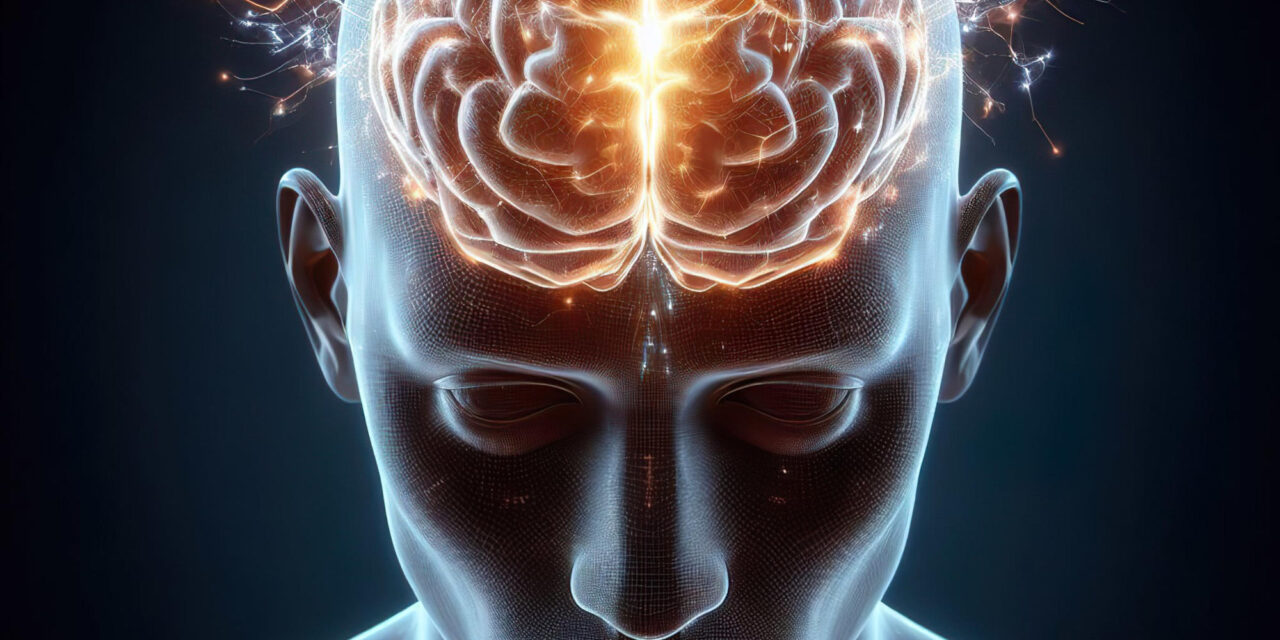
Lung cancer cells have been discovered to form functional synaptic connections with neurons in the brain, a devious mechanism that allows the cancer to hijack neural communication pathways to spur tumor growth and progression. This breakthrough finding, published recently in Nature, reveals how small cell lung cancer (SCLC) cells synapse with neurons, receiving chemical signals that trigger electrical changes and calcium surges within the cancer cells, fueling their proliferation and brain metastasis. This connection explains the rapid progression and poor prognosis of lung cancer brain metastases, especially in SCLC, which accounts for 15% of all lung cancers but has a median survival of just 12 months once brain metastases develop. About 60% of these patients already have brain metastases at diagnosis, making this discovery crucial for understanding and potentially targeting the deadly spread of lung cancer to the brain.
Key Research Findings on Lung-Brain Cancer Connection
Two independent research teams, one from Stanford Medicine and another in Germany, explored how lung cancer cells establish neural synapses in the brain. They found that SCLC cells mimic neurons by forming complete synaptic structures, including postsynaptic densities and neurotransmitter vesicles that enable electrochemical signaling via glutamate and gamma-aminobutyric acid (GABA). This neural hijacking allows lung cancer cells to integrate into brain circuits usually reserved for learning and memory processes, fundamentally changing how the cancer thrives in the brain environment. Electron microscopy confirmed these direct neuron-to-cancer cell synapses, highlighting that lung cancer cells bring their own neural wiring to the brain, contributing to aggressive tumor growth and progression despite conventional therapies like chemotherapy and immunotherapy.
Expert Commentary and Public Health Implications
Experts highlight the significance of this discovery, noting it as a paradigm shift in understanding brain metastases from lung cancer. Dr. Michelle Monje, a neuro-oncologist who led part of the study, expressed surprise and concern about the aggressive nature of SCLC’s neural integration. This finding opens avenues for new treatment strategies that could disrupt these synaptic connections or block the neural signaling that promotes tumor growth. Given that brain metastases severely impact patient quality of life and survival, with neurological symptoms such as headaches, cognitive impairments, and seizures common, targeting these pathways could improve outcomes. Additionally, this research underscores the urgent need for early detection and management of brain metastases in lung cancer patients, especially since many already have brain involvement at diagnosis.
Background and Context of Lung Cancer Brain Metastasis
Brain metastases are a frequent and severe complication of lung cancer, especially small cell lung cancer and certain subtypes of non-small cell lung cancer (NSCLC) such as EGFR-mutated or ALK-rearranged forms. Up to 50% of lung cancer patients develop brain metastases during their disease course, contributing to high mortality. Current treatments include chemotherapy, radiation therapies, and newer targeted therapies, but the blood-brain barrier limits drug effectiveness. Brain metastases often manifest as multiple lesions and cause significant neurological deficits, further complicating management. Advances in imaging techniques, including MRI, aid early detection, yet survival remains poor due to the aggressive biology of brain metastases and the challenges in treating cancer cells that effectively “plug into” brain circuits.
Limitations and Future Directions
While this discovery adds critical understanding to lung cancer brain metastasis biology, challenges remain. The studies primarily used preclinical models, and more clinical trials are needed to assess therapies targeting the synaptic interactions. There are also conflicts regarding the heterogeneity of tumors and the varying mechanisms that different lung cancer subtypes use to metastasize. Additionally, the complexity of brain tumor microenvironments and the blood-brain barrier pose obstacles to effective drug delivery. Future research aims to develop treatments that can disrupt neuron-cancer synapses or modulate neurotransmitter pathways like glutamate signaling to inhibit tumor growth. Furthermore, interdisciplinary approaches integrating neurobiology, oncology, and pharmacology will be paramount to translate these findings into clinical solutions.
Practical Implications for Readers
For the general public, especially those affected by lung cancer, this research emphasizes the importance of vigilant neurological monitoring and early diagnostic imaging for brain metastases. Understanding the aggressive nature of brain spread helps patients and caregivers appreciate the complexities in treatment decisions. While new targeted therapies and immunotherapies are evolving, ongoing research like this aims to provide more effective, personalized options in the near future. Maintaining communication with healthcare providers about symptoms such as headaches, cognitive changes, or neurological deficits is vital. This discovery is a step towards more hopeful interventions for one of the deadliest lung cancer complications, potentially improving patient survival and quality of life.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
-
Venkatesh, H. S., Monje, M., Beleggia, F. et al. (2025). Small cell lung cancer forms functional synaptic connections with neurons to drive brain metastasis. Nature. DOI: 10.1038/s41586-025-09492-z.medscape+1