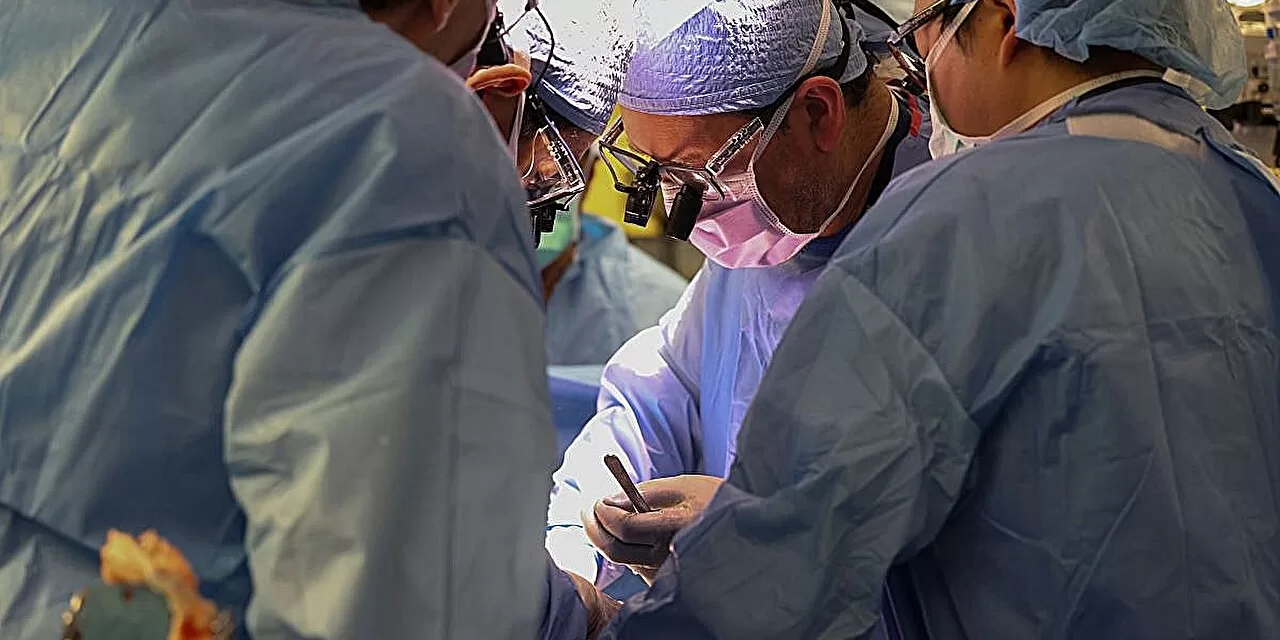
In a pioneering medical feat reported in Nature Medicine, a team of surgeons in China successfully implanted a gene-edited pig lung into a human recipient for the first time, marking a critical advancement in xenotransplantation. The lung functioned for nine days, exchanging oxygen effectively before immune responses led to tissue damage. This groundbreaking procedure addresses the urgent global shortage of donor organs and provides valuable insights into overcoming barriers in cross-species transplantation.
Key Findings and Medical Breakthrough
In this landmark case, surgeons at the First Affiliated Hospital of Guangzhou Medical University connected a gene-edited lung from a Bama Xiang pig to a 39-year-old man who had been declared brain dead following a severe brain bleed. The transplanted lung successfully exchanged oxygen for 216 hours (nine days), a crucial demonstration of basic lung function across species barriers. This work represents an unprecedented step forward in overcoming the critical shortage of donor lungs, for which thousands of patients worldwide are currently waiting. In 2024, for instance, the United States performed only 3,340 lung transplants, leaving many patients on long, often fatal waiting lists .
The pig lung underwent gene editing through CRISPR technology, where three pig genes known to provoke strong immune rejection were deleted, and three human genes were inserted to modulate blood clotting and complement activation pathways. This strategic genetic modification alongside immunosuppressive drug therapy prevented hyperacute (immediate) rejection—a major obstacle that historically prevented the success of xenotransplantation .
Expert Commentary and Perspectives
Dr. Jianxing He, the lead surgeon, highlighted the significance of this trial as a vital learning platform. “This experiment offers a detailed timeline of immune reactions in a pig lung connected to human circulation, providing a foundation for future improvements,” he stated. Experts outside the study underscore its importance as well: Richard Pierson III, a transplant surgeon at Massachusetts General Hospital, described the work as “exciting and an essential building block for incremental progress in xenotransplantation.” Similarly, Justin Chan from NYU Langone emphasized the necessity of cautious, data-driven trials before moving to living recipients .
Context and Background on Xenotransplantation
Xenotransplantation—the transplantation of animal organs into humans—has long been explored as a solution to the global shortage of donor organs. While pig kidneys have recently demonstrated promising outcomes in living recipients lasting months, lungs present unique challenges due to their constant exposure to air, microbes, and a dense immune cell network. This environment predisposes lungs to intense immune responses and inflammation, often leading to early graft failure even in human-to-human transplants. The lung’s fragile nature makes xenotransplantation especially difficult, requiring more sophisticated genetic and pharmacological approaches than those applied to kidneys or livers .
Implications for Public Health and Patients
The success of this initial trial could pave the way for future therapies for patients with severe lung diseases such as cystic fibrosis, pulmonary fibrosis, or emphysema, many of whom currently have very limited treatment options. If researchers can refine gene editing and improve immunosuppressive regimens, pig lungs might become a viable alternative to human donor lungs, potentially saving thousands of lives.
Moreover, expanded understanding of immune rejection timing and the inflammatory cascade will inform clinical protocols and pharmaceutical development, enhancing survival rates post-transplant. The study also encourages innovation in organ preservation and perfusion techniques to minimize early graft injury—a critical factor for transplantation success .
Potential Limitations and Counterarguments
Despite these promising findings, several challenges remain. The nine-day duration, while groundbreaking, is relatively short and conducted in a brain-dead individual whose condition itself induces high systemic inflammation, complicating interpretation. The transplanted pig lung was tested alongside the patient’s native lung, hampering clear assessment of full organ function in isolation.
Furthermore, later immune responses included antibody binding and complement activation that caused swelling, inflammation, and reduced lung compliance—signs of ongoing rejection that must be addressed for long-term success. Experts stress the need for cautious optimism and further research to ensure safety and efficacy in living recipients .
Conclusion
This successful implantation of a gene-edited pig lung into a human marks a critical milestone in medical history, opening new avenues for addressing organ shortages through xenotransplantation. While the journey to routine clinical use remains complex, this study lays important groundwork through its detailed monitoring of immune interactions over nine days. The promise of gene editing combined with evolving immunosuppressive therapies gives hope of transforming lung transplantation and saving countless lives worldwide.
Medical Disclaimer
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
- https://www.earth.com/news/doctors-successfully-implant-a-pigs-lung-into-a-human-for-the-first-time/