Scientists have recently developed an innovative automated tactile stimulation device named Breathing Operator for BaBY (BOBBY) designed to aid preterm infants who struggle with independent breathing. This device aims to provide immediate, consistent, and gentle stimulation mimicking neonatal nurses’ manual touch to prompt breathing during respiratory pauses, potentially transforming neonatal intensive care.
Addressing Breathing Challenges in Preterm Infants
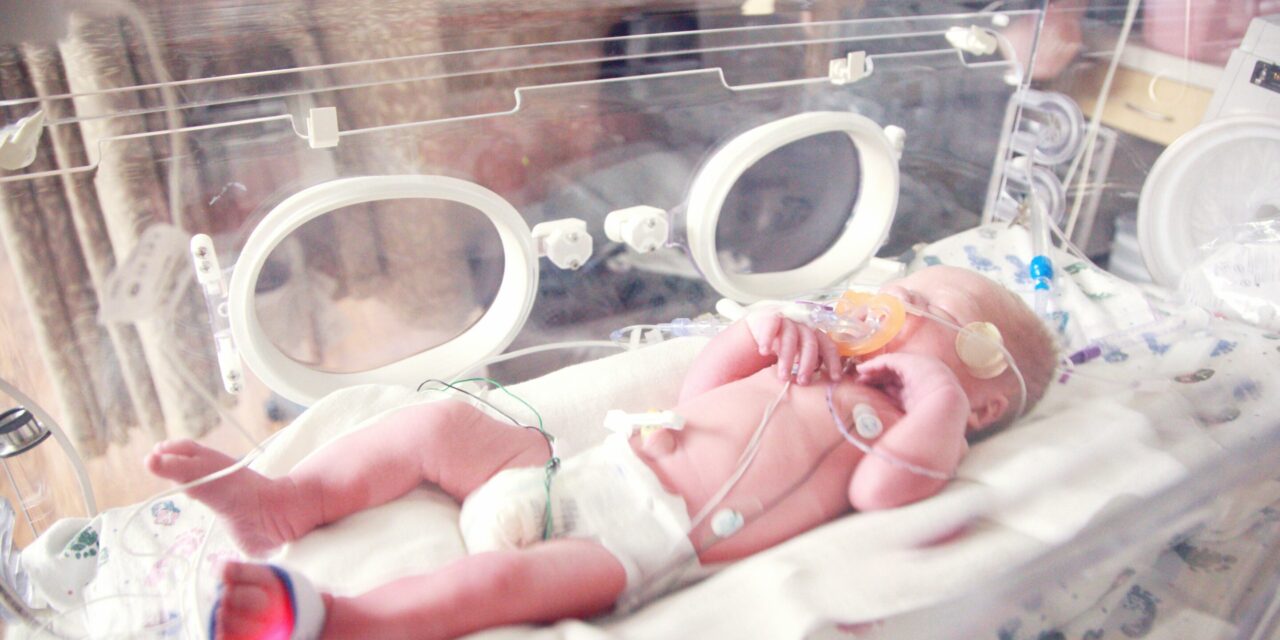
Preterm infants, especially those born before 30 weeks gestation, often experience immature lungs and underdeveloped respiratory control systems. This immaturity leads to frequent breathing pauses or apnea, which can quickly cause drops in oxygen levels and result in brain damage or developmental delays if prolonged. Traditionally, neonatal nurses respond by manually applying tactile stimulation—rubbing the infant’s back or soles of feet—to trigger resumption of breathing. However, this manual method is often inconsistent and dependent on nurse availability, risking delayed response during critical moments.
The BOBBY device was developed to address these challenges by automating this tactile stimulation as soon as cardiorespiratory monitoring alarms detect a breathing pause. By providing timely intervention without delay, the device aims to reduce the risks associated with apnea and lessen the workload and alarm fatigue experienced by neonatal nurses.
Innovative Design and Features of BOBBY
The development of BOBBY followed a rigorous five-phase iterative design process involving discovery, definition, ideation, prototyping, and refinement. Scientists conducted detailed studies on the clinical context and needs in neonatal intensive care units (NICUs) and collaborated extensively with neonatal specialists and nurses to ensure the device meets real-world requirements.
A defining feature of BOBBY is its use of “apparent tactile motion” technology, which replicates the gentle, rhythmic touch typically used by nurses to prompt breathing. This approach balances efficacy with a safety profile that minimizes the risk of skin irritation. Design considerations ensure that the electronic components do not directly contact the infant, adhering to strict safety standards.
Clinical Feasibility and Expert Insights
Initial feasibility studies show promising results. In a randomized cross-over trial involving 16 preterm infants, BOBBY successfully detected 84% of cardiorespiratory events and automatically applied stimulation in response 100% of the time. Nurses found the device user-friendly and clinically useful in managing respiratory pauses. However, one infant did develop mild skin erythema leading to device removal, underscoring the need for continued safety evaluations.
Sophie Cramer, a medical engineer involved in the project, highlights the device’s potential to act as “an extra pair of hands,” delivering immediate support during apnea episodes while nurses focus on other critical care aspects. She emphasizes that while manual techniques have historically been effective, they vary widely and lack scientific standardization—a gap BOBBY aims to fill with consistent, evidence-based intervention.
Neonatology experts not involved in the development echo similar views. Dr. Anita Sharma, a neonatologist at a major children’s hospital, explains that automated tactile stimulation can reduce response time delays that often occur due to alarm fatigue. “Any tool that reliably reduces the duration of apnea can significantly improve neurological outcomes in these vulnerable infants,” she notes .
Potential Impact on Public Health and Neonatal Care
The introduction of BOBBY in NICUs could alleviate the high workload and stress on nursing staff by automating urgent interventions. This may also enhance respiratory outcomes and reduce complications related to apnea, thereby improving survival rates and long-term health in preterm populations.
It is important to place BOBBY in the context of existing respiratory support methods such as continuous positive airway pressure (CPAP), caffeine therapy, and invasive ventilation. These interventions focus on lung support and stimulation of the respiratory drive but may be complemented by timely tactile stimulation to address acute apnea episodes.
Limitations and Ongoing Research
Researchers acknowledge that while early results are encouraging, BOBBY’s widespread adoption awaits further robust clinical trials to confirm safety, efficacy, and long-term benefits. Potential limitations include skin irritation risks, the device’s sensitivity and specificity in detecting apnea, and integration with existing neonatal monitoring systems.
Moreover, BOBBY is designed as a supplementary tool rather than a replacement for nursing care. Careful protocols will be necessary to ensure the device augments rather than diminishes human vigilance, especially in complex neonatal cases.
Practical Implications for Families and Care Providers
For families of preterm infants, the availability of an automated stimulation device could mean enhanced safety and reduced risks related to apnea. For healthcare professionals, BOBBY offers a precision, evidence-based tool to support breathing, potentially standardizing interventions and improving workflows in busy NICUs.
Parents and caregivers should understand that this device is part of a comprehensive neonatal care approach, which includes respiratory support, medication, and vigilant monitoring by healthcare staff .
Medical Disclaimer
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.