Hyderabad, May 22, 2025:
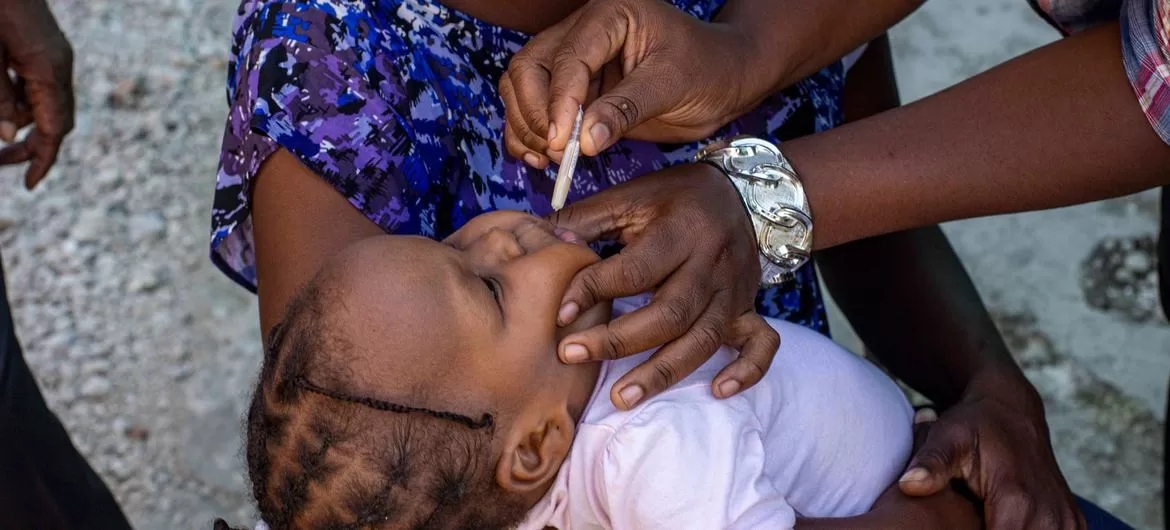
Bharat Biotech, a leading vaccine manufacturer based in Hyderabad, has announced the successful completion of Phase III clinical trials for its new oral cholera vaccine, Hillchol. The results mark a significant milestone in the fight against cholera, a disease that continues to pose a major global health threat, especially in lower- and middle-income countries.
Strong Efficacy and Safety Profile
The Phase III clinical trial, which was double-blind and randomized, evaluated the safety, immunogenicity, and efficacy of Hillchol in comparison to an existing licensed vaccine, Shanchol. The study involved 1,800 participants, ranging from infants to adults, across 10 clinical sites in India. The participants were divided into three age groups: adults (over 18 years), children (5 to under 18 years), and infants (1 to under 5 years).
Hillchol demonstrated robust efficacy, showing an over four-fold rise in vibriocidal antibodies—68.3% for the Ogawa serotype and 69.5% for the Inaba serotype of the cholera bacterium. The vaccine was found to be non-inferior to the comparator vaccine and maintained a strong safety profile across all age groups, with only mild adverse events reported.
Addressing Global Vaccine Shortages
Dr. Krishna Ella, Executive Chairman of Bharat Biotech, highlighted the importance of Hillchol in addressing the ongoing global cholera crisis and vaccine shortages. The new-generation vaccine features a simplified, single stable O1 Hikojima strain, which enhances production efficiency and affordability. This is expected to be particularly beneficial for countries with limited resources.
Bharat Biotech’s manufacturing facilities in Hyderabad and Bhubaneswar are equipped to produce up to 200 million doses of Hillchol annually. This capacity could help alleviate the global shortage of oral cholera vaccines, as current global demand stands at around 100 million doses per year, with only one manufacturer currently supplying the market.
The Ongoing Fight Against Cholera
Cholera, caused by ingesting food or water contaminated with the Vibrio cholerae bacteria, is estimated to cause over 2.86 million cases and approximately 95,000 deaths worldwide each year. The development of new, effective, and affordable vaccines like Hillchol is crucial in reducing the global burden of this potentially fatal disease.
Bharat Biotech has a strong track record in vaccine innovation, having delivered over nine billion doses globally and holding over 145 patents. The company’s portfolio includes vaccines for Covid-19 (Covaxin), influenza H1N1, rotavirus, Japanese encephalitis, rabies, chikungunya, Zika, and typhoid.
What’s Next?
The findings from the Phase III trial have been published in the ScienceDirect vaccine journal. With regulatory approvals pending, Hillchol could soon become a key tool in global cholera prevention efforts.
Disclaimer:
This article is based on information reported by The Times of India on May 22, 2025, regarding Bharat Biotech’s announcement of successful Phase III trials for the Hillchol oral cholera vaccine. The vaccine is not yet commercially available and awaits regulatory approval. For medical advice or vaccination recommendations, please consult a healthcare professional.
Citations: