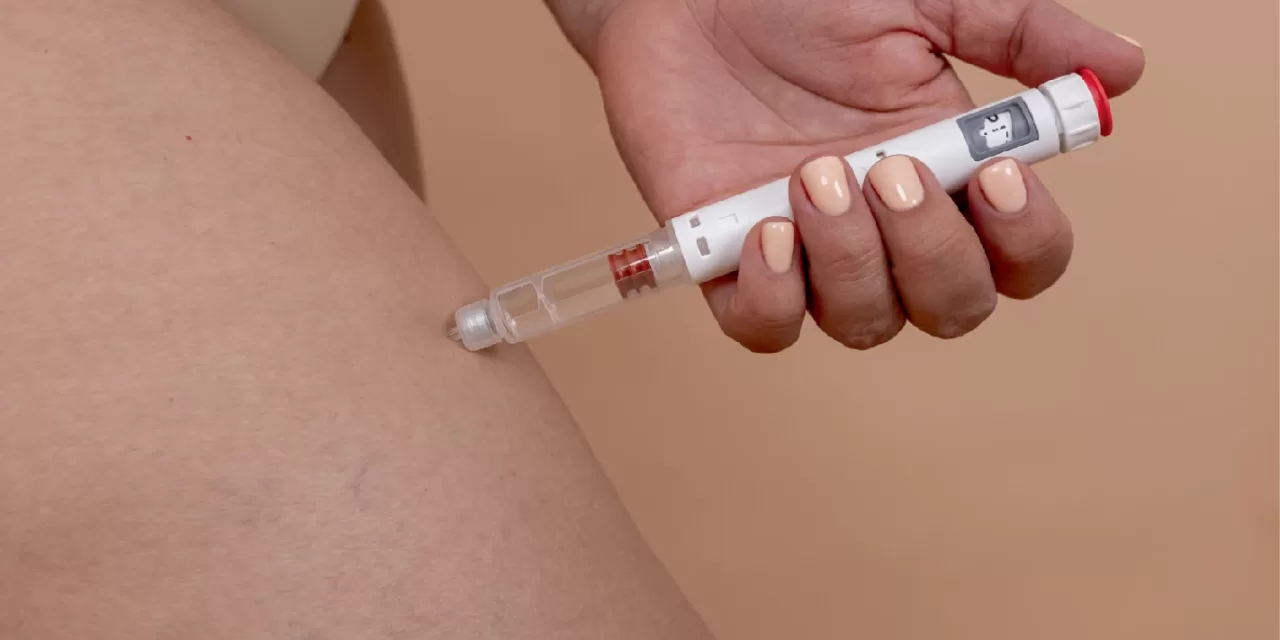
MUMBAI – A high-stakes pharmaceutical showdown is unfolding in India as the world’s two largest producers of weight-loss medications, Eli Lilly and Novo Nordisk, accelerate their efforts to capture a dominant share of the country’s rapidly expanding obesity market.
This week, industry reports and regulatory filings indicate that both companies are aggressively navigating India’s complex healthcare landscape to launch their blockbuster GLP-1 receptor agonists—Zepbound (tirzepatide) and Wegovy (semaglutide). The moves come at a critical time for India, which is grappling with a dual burden of malnutrition and a rising tide of metabolic disorders, including type 2 diabetes and obesity.
The Scale of the Crisis
For decades, public health efforts in India focused primarily on infectious diseases and undernutrition. However, rapid urbanization and shifting dietary patterns have led to a “globesity” crisis. According to the World Obesity Federation’s 2023 Atlas, India is projected to see an annual increase in adult obesity of 5.2% between 2020 and 2035.
Currently, estimates from the Indian Council of Medical Research (ICMR-INDIAB study) suggest that over 100 million Indians are living with diabetes, while more than 135 million are classified as obese or overweight. This demographic represents a massive, untapped market for pharmaceutical giants.
“We are seeing a tectonic shift in the metabolic health of our population,” says Dr. Anirban Biswas, an endocrinologist based in New Delhi, who is not affiliated with either company. “For many of our patients, lifestyle modifications alone aren’t stemming the tide of complications like heart disease and fatty liver. The entry of these advanced therapies is not just a commercial race; it’s a public health necessity.”
The Contenders: Tirzepatide vs. Semaglutide
The competition centers on two highly effective, injectable medications that mimic hormones in the gut to reduce appetite and slow digestion.
-
Novo Nordisk’s Wegovy (Semaglutide): Already a household name in the West, Wegovy has demonstrated an average weight loss of approximately 15% in clinical trials over 68 weeks. Novo Nordisk has a long-standing presence in India through its insulin portfolio, giving it a robust distribution network.
-
Eli Lilly’s Zepbound/Mounjaro (Tirzepatide): Lilly’s contender is a “dual agonist,” targeting both GLP-1 and GIP receptors. Clinical data published in The New England Journal of Medicine showed that participants on the highest dose of tirzepatide lost an average of 20.9% of their body weight over 72 weeks, potentially giving it an edge in efficacy.
While Novo Nordisk is leveraging its established supply chain, Eli Lilly has recently sought to clear regulatory hurdles for a direct-to-consumer launch, aiming to bypass middle-market delays that have historically slowed the introduction of Western drugs in India.
Challenges: Pricing and Accessibility
While the medical potential is significant, the primary barrier in the Indian market is affordability. Unlike in many European nations, healthcare in India is largely an out-of-pocket expense for the middle class.
The projected cost for a monthly supply of these medications is expected to range between ₹15,000 and ₹30,000 ($180–$360 USD). In a country where the average monthly income is significantly lower, these drugs risk becoming “boutique” treatments for the urban elite rather than tools for broad public health.
“The challenge for Lilly and Novo is not just getting the drugs onto shelves, but making them sustainable for the Indian economy,” explains Sarthak Sen, a pharmaceutical industry analyst. “Both companies are reportedly exploring tiered pricing models and local manufacturing partnerships to reduce costs and satisfy the Indian government’s ‘Make in India’ requirements.”
Cultural and Medical Considerations
Beyond the numbers, medical professionals emphasize that these drugs are not “magic bullets.” In the Indian context, where muscle mass tends to be lower and visceral fat (fat around the organs) tends to be higher at lower BMIs, the administration of GLP-1s requires careful supervision.
“We must be cautious about the ‘quick fix’ narrative,” warns Dr. Priya Venkatesh, a metabolic specialist. “These are chronic medications. If a patient stops the drug without significant lifestyle changes, the weight often returns. Furthermore, we need more data on how these drugs perform specifically within South Asian genotypes, who are predisposed to insulin resistance at lower body weights.”
Common side effects reported in global trials include nausea, vomiting, and diarrhea. There are also rare but serious risks, such as pancreatitis and gallbladder problems, which necessitate rigorous medical screening before a prescription is issued.
The Road Ahead
The Indian government’s drug regulator, the Central Drugs Standard Control Organisation (CDSCO), is currently reviewing data from local bridging trials. These trials are essential to ensure the drugs are safe and effective for the Indian population.
Industry insiders suggest that 2025 will be the “Year of the GLP-1” in India, with both companies expected to have full-scale commercial launches by the second half of the year. The outcome of this race will likely dictate the landscape of metabolic healthcare in South Asia for the next decade.
For the millions of Indians struggling with obesity-related comorbidities, the competition between Eli Lilly and Novo Nordisk offers a glimmer of hope—provided the price is right and the medical oversight is sound.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
-
Reuters: “Lilly, Novo Nordisk lock horns in India’s obesity drug race” (Dec 25, 2025).