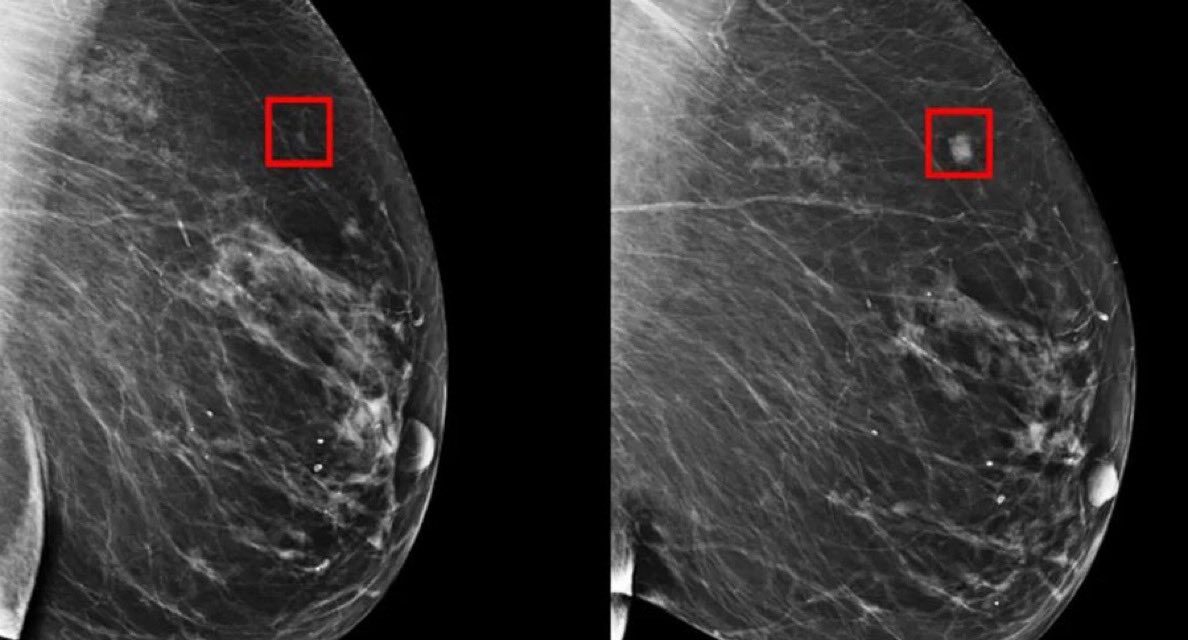
In a major advance reported on October 18, 2025, AstraZeneca and Daiichi Sankyo announced compelling results from two pivotal clinical trials of their drug Enhertu in early-stage HER2-positive breast cancer. Presented at the European Society for Medical Oncology Congress, the findings indicate that Enhertu, already approved for more advanced cancer stages, may become a new standard of care for high-risk early breast cancer, potentially improving disease outcomes dramatically for patients worldwide.
Enhertu is an antibody-drug conjugate (ADC) designed to precisely target HER2-positive cancer cells while sparing healthy tissue. HER2-positive breast cancer accounts for approximately 15-20% of breast cancer cases. The drug is often described as a “guided missile” therapy due to its ability to deliver chemotherapy directly to cancer cells, reducing damage to normal cells compared to traditional chemotherapy.
Key Findings and Clinical Outcomes
One late-phase, randomized trial compared Enhertu to Roche’s Kadcyla in patients with high-risk early-stage HER2-positive breast cancer that persisted after surgery and initial treatment. The study revealed a 53% reduction in the risk of invasive disease recurrence or death with Enhertu. After three years, 92.4% of patients treated with Enhertu remained alive and disease-free, compared to 83.7% of those receiving Kadcyla. This translates to a significant improvement in long-term survival and disease prevention.
In a second trial, Enhertu was administered prior to surgery (neoadjuvant setting) and followed by standard chemotherapy (THP regimen). The pathologic complete response (pCR) rate—meaning no evidence of invasive cancer in breast tissue after treatment—was 67.3% with Enhertu, surpassing the 56% pCR rate seen with chemotherapy alone. Importantly, patients also experienced fewer severe adverse events, indicating improved tolerability.
Expert Insight
Dr. Sarah Galbra, AstraZeneca’s Executive Vice President for Oncology Research and Development, highlighted the significance: “These results validate our vision for antibody-drug conjugates to increasingly replace chemotherapy in various treatment contexts, offering patients better outcomes with less toxicity.”
Ken Keller, Global Head of Oncology at Daiichi Sankyo, emphasized their strategic approach: “Wherever chemotherapy is effective, we explore if our DXd ADCs can deliver superior results. The evidence from these studies strengthens that case.”
Current Use and Future Directions
Enhertu’s success builds on its prior approvals for advanced breast cancer and other cancer types, including lung and gastric cancers. In 2024, Enhertu generated combined revenues of $3.75 billion for AstraZeneca and Daiichi Sankyo and is authorized in over 75 countries. These new findings pave the way for regulatory submissions to expand its use to earlier stages of HER2-positive breast cancer, potentially offering a curative option for patients at high risk of recurrence.
Implications for Public Health and Patients
For patients diagnosed with early-stage HER2-positive breast cancer, these data represent hope for more effective treatments that can prevent recurrence and improve survival with manageable side effects. The higher pathologic complete response rates suggest potential for less invasive surgery and better long-term prognosis.
The ADC technology underpinning Enhertu exemplifies precision medicine’s advance in oncology: treatments that specifically target cancer cells while limiting harm to normal tissues, reducing chemotherapy’s notorious side effects.
Limitations and Balanced Considerations
While promising, these results come from controlled clinical trial settings. Long-term real-world effectiveness and safety data are needed. Additionally, access and affordability remain challenges, particularly in lower-resource settings. Experts note that regulatory review timelines and reimbursement issues will influence how quickly patients worldwide can benefit.
As with any novel therapy, ongoing monitoring for rare adverse effects and confirmation of long-term outcomes will be critical. Further research will clarify the optimal patient subgroups and combination regimens to maximize benefits.
Conclusion
Enhertu’s promising clinical trial results mark a significant step forward in treating early-stage HER2-positive breast cancer, offering patients a potential new standard of care that integrates precision-targeted therapy beyond traditional chemotherapy. This development exemplifies progress in oncology towards more effective, tailored treatments to improve survival and quality of life.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References