A groundbreaking approach to personalized cancer treatment is being pioneered by a team of researchers from the University of Duisburg-Essen (UDE), LMU Munich, and the Berlin Institute for the Foundations of Learning and Data (BIFOLD) at TU Berlin. Their innovative use of artificial intelligence (AI) promises to revolutionize how cancer treatments are tailored to individual patients.
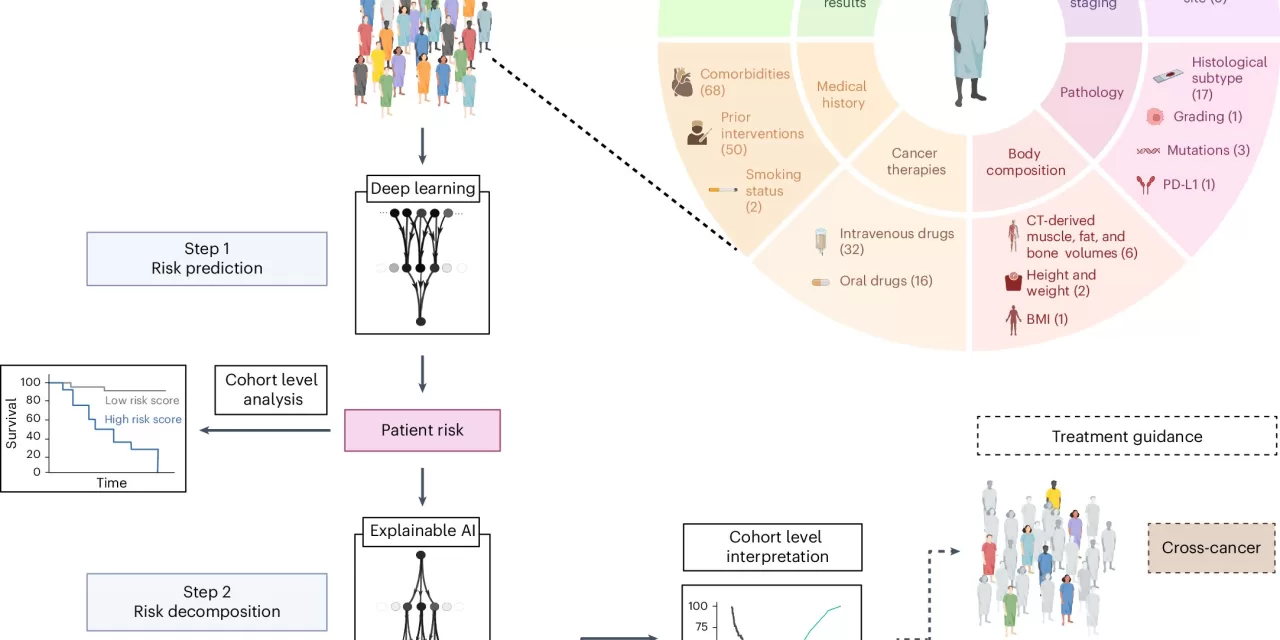
Traditionally, personalized medicine has relied on a small set of parameters, such as age, gender, and genetic information, to predict the course of a disease. However, these parameters are often insufficient to fully grasp the complexities of diseases like cancer. The researchers at UDE, LMU, and BIFOLD have taken a major step forward by integrating vast amounts of clinical data from different sources, including medical history, laboratory values, imaging, and genetic analyses, to support clinical decision-making.
The team’s work builds on the smart hospital infrastructure at University Hospital Essen, where a cutting-edge AI model was trained using data from over 15,000 cancer patients with 38 different types of solid tumors. This AI model examines the interaction of 350 different parameters, revealing key prognostic factors and uncovering complex interrelationships that could otherwise go unnoticed.
“Although large amounts of clinical data are available in modern medicine, the promise of truly personalized medicine often remains unfulfilled,” said Prof. Jens Kleesiek from the Institute for Artificial Intelligence in Medicine at University Hospital Essen. This new AI-driven approach, however, promises to offer more than just incremental improvements—it could dramatically enhance the way clinicians understand and treat cancer.
The researchers focused on the use of explainable artificial intelligence (xAI), a technology that not only deciphers complex interrelationships between various data points but also provides transparent, comprehensible decisions. With this approach, clinicians can understand exactly how each parameter contributed to the AI’s prognosis for a patient, ensuring that treatment plans are based on more than just raw data.
For their study, published in Nature Cancer, the team examined how 350 parameters interact to influence cancer treatment outcomes. These parameters included clinical data, laboratory values, imaging results, and genetic tumor profiles. By doing so, the AI was able to identify key factors that influence the decision-making process in cancer treatment, highlighting important, previously undetected interactions.
In clinical trials, the AI model demonstrated its accuracy by being tested on data from more than 3,000 lung cancer patients. The AI’s ability to integrate diverse data sources and make transparent, personalized predictions could significantly improve patient outcomes.
“Our results show the potential of artificial intelligence to look at clinical data not in isolation but in context, to re-evaluate them, and thus enable personalized, data-driven cancer therapy,” said Dr. Philipp Keyl from LMU. The technology may also be beneficial in emergency situations, where rapid assessment of diagnostic parameters is critical.
Looking ahead, the team aims to explore cross-cancer interrelationships that were previously hidden using conventional statistical methods. Through collaboration with the National Center for Tumor Diseases (NCT) and other oncological networks, they are preparing for the next phase of their research: proving the real-world benefits of this AI model in clinical trials.
As Prof. Martin Schuler, Managing Director of the NCT West site, emphasized, “We have the ideal conditions to take the next step: proving the real patient benefit of our technology in clinical trials.”
This research is a significant leap toward making personalized cancer treatments more effective and accessible to patients worldwide.
Disclaimer: This article is based on research published in Nature Cancer (2025), and the claims made in this study should be understood within the context of ongoing research and clinical trials. The findings are not yet proven in routine clinical practice, and further validation is required before widespread adoption.
For more information, please refer to: Julius Keyl et al, Decoding pan-cancer treatment outcomes using multimodal real-world data and explainable artificial intelligence, Nature Cancer (2025). DOI: 10.1038/s43018-024-00891-1