SHANGHAI — In a landmark decision that shifts the global landscape of neurotechnology, China’s National Medical Products Administration (NMPA) announced on March 13, 2026, the market approval of the world’s first invasive brain-computer interface (BCI) medical device. Developed by Shanghai-based Neuracle Medical Technology Co., Ltd. (also known as Borui Kang), the implantable system is designed to restore hand-grasping function in adults living with paralysis due to cervical spinal cord injuries.
This regulatory milestone represents a significant leap from laboratory research to commercial availability. While high-profile competitors like Elon Musk’s Neuralink continue to navigate human clinical trials in the United States, China’s approval marks the first time such an invasive neural implant has been cleared for widespread clinical use, positioning the nation at the forefront of a burgeoning “future industry.”
Precision Through Procedure: How the Device Works
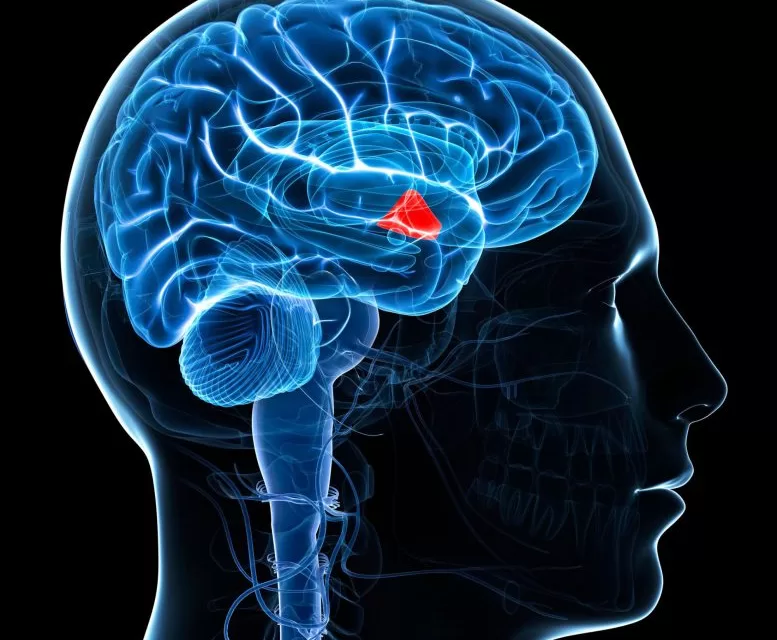
The Neuracle BCI system is a sophisticated bridge between the human brain and the physical world. Unlike non-invasive BCIs, which use external sensors placed on the scalp and often struggle with signal “noise,” this system utilizes an electrode array placed extradurally—positioned just above the brain’s outermost protective membrane (the dura mater).
This minimally invasive approach is a strategic middle ground: it provides the high-fidelity signal precision necessary for complex motor control while avoiding the risks associated with penetrating the brain tissue itself. The system works through a four-stage process:
-
Capture: Wireless electrodes detect neural signals related to intent.
-
Transmission: A neural signal transmitter sends these pulses to an external receiver.
-
Decoding: Advanced software translates brain activity into digital commands.
-
Action: A specialized pneumatic glove on the patient’s hand executes the physical grasping motion.
The device is specifically indicated for adults aged 18 to 60 who have sustained C2-C6 cervical spinal cord injuries. Eligible patients include those with Grade A through C injuries (ranging from complete to incomplete paralysis) who have been stable for at least six months but lack functional use of their hands.
Clinical Breakthroughs and Remodeling
The NMPA’s approval followed rigorous testing, including 36 procedures across four feasibility studies and 32 multi-center Good Clinical Practice (GCP) trials. The results were compelling: all participants demonstrated improved grasping capabilities.
Perhaps most significantly, researchers observed signs of neural remodeling in some participants. This suggests that the consistent use of the BCI might help the brain “rewire” or strengthen existing neural pathways, potentially offering benefits that extend beyond the device’s mechanical assistance.
“This approval marks a pivotal moment for BCI translation from lab to clinic, potentially accelerating global adoption,” says Dr. Li Wei, a neuroengineer at Tsinghua University’s Institute for Brain Research. Dr. Li, who was not involved in the trials, noted that China’s streamlined regulatory path for high-priority technology could see BCI use become a standard rehabilitation option within three to five years.
The Global Neurotech Race
The commercialization of the Neuracle device underscores a shifting dynamic in the global “brain race.” For decades, BCI technology was largely experimental, but China has now designated it a “future industry” in its 2026 government work report.
| Aspect | China’s Neuracle BCI | Neuralink (U.S.) |
| Approval Status | Market-approved (March 2026) | Human trials ongoing |
| Primary Target | Hand grasp / Spinal injury | Broader motor/sensory restoration |
| Implantation | Minimally invasive extradural | Fully invasive “threads” |
| Human Data | 36 human procedures | ~10 humans (estimated) |
While Neuralink aims for a broader range of applications—including vision restoration and high-bandwidth data transfer—China’s strategy appears focused on immediate, functional relief for paralysis patients. Experts predict the Chinese BCI market could reach $530 million by the end of 2025, driven by government backing and expanding insurance coverage.
Public Health and Daily Life
The implications for public health are profound. Globally, between 250,000 and 500,000 people suffer spinal cord injuries annually. The lifetime cost for a single patient in high-income countries can exceed $1 million, largely due to the need for 24-hour care.
By restoring hand function, the Neuracle device aims to:
-
Enhance Autonomy: Allow patients to perform daily tasks like eating, dressing, and using mobile devices without assistance.
-
Reduce Caregiver Burden: Decrease the level of constant physical support required from family members or medical staff.
-
Improve Mental Health: Combat the social isolation and depression often associated with total loss of limb function.
Safety Considerations and Ethical Hurdles
Despite the optimism, the medical community remains cautious. Dr. Miguel Nicolelis, a pioneer in BCI research at Duke University, has previously noted that invasive implants face long-term hurdles. “Signal degradation over time and immune responses are common hurdles,” he emphasized in discussions regarding similar technologies.
Potential risks include:
-
Infection: Standard for any implantable device, typically ranging from 1% to 5% in similar neurosurgical procedures.
-
Mechanical Integrity: Issues such as electrode migration or battery life limitations.
-
Signal Drift: The need for frequent software recalibration as the brain or the implant site changes over time.
Ethical concerns also loom large. As BCI devices move into the commercial sector, questions regarding neural data privacy—who owns the signals recorded from a person’s brain—and equitable access for patients in low-resource settings remain unanswered.
Looking Ahead
The approval of the Neuracle BCI is not a “cure” for paralysis, but rather a powerful adjunctive tool in the rehabilitation toolkit. For the hundreds of thousands of individuals living with spinal cord injuries, it represents a bridge to a more independent life. As the first units reach the market, the global medical community will be watching closely to see if the real-world performance matches the promise of the clinical trials.
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.
References
- https://www.reuters.com/business/healthcare-pharmaceuticals/china-approves-market-launch-brain-computer-interface-medical-device-world-first-2026-03-13/