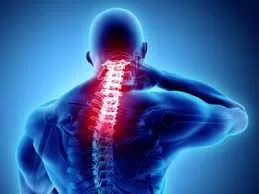
In a groundbreaking development, results from a phase 1 clinical trial conducted by the Mayo Clinic in the United States suggest that stem cell therapy could offer a safe and potentially effective treatment option for individuals grappling with hard-to-treat traumatic spinal cord injuries. The trial, published on Monday by the Mayo Clinic, unveils promising outcomes indicating that stem cells derived from a patient’s own fat may not only be safe but could also contribute to improvements in sensation and movement for those living with spinal cord injuries and paralysis.
Mohamad Bydon, a neurosurgeon at Mayo Clinic, expressed optimism about the study’s findings, stating, “This study documents the safety and potential benefit of stem cells and regenerative medicine.”
Published in the journal Nature Communications, the trial involved 10 adults, seven of whom exhibited notable improvements following stem cell therapy. These improvements included increased sensation upon testing with pinprick and light touch, enhanced strength in muscle motor groups, and even recovery of voluntary anal contraction, crucial for bowel function.
Although three patients did not demonstrate improvement, their condition did not deteriorate either, underscoring the overall safety of the treatment. Notably, no serious adverse events were recorded post-treatment, with the most commonly reported side effects being headache and musculoskeletal pain, which were effectively managed with over-the-counter treatments.
Dr. Bydon emphasized the significance of even modest improvements, stating, “Even a mild improvement can make a significant difference in that patient’s quality of life.”
The World Health Organization (WHO) estimates that between 250,000 and 500,000 individuals worldwide sustain spinal cord injuries annually, yet treatment options remain profoundly limited. Historically, supportive care, stabilization surgery, and physical therapy have constituted the primary modalities for managing spinal cord injuries.
However, Dr. Bydon highlighted the potential for stem cell therapy to herald a new era in the treatment of spinal cord injuries. He noted, “Spinal cord injury is a complex condition. Future research may show whether stem cells in combination with other therapies could be part of a new paradigm of treatment to improve outcomes for patients.”
The promising results from this phase 1 clinical trial offer hope for individuals grappling with traumatic spinal cord injuries, paving the way for further research and potential advancements in regenerative medicine to transform the landscape of spinal cord injury treatment.
This pioneering study underscores the Mayo Clinic’s commitment to advancing medical innovation and underscores the profound impact that stem cell therapy could have on improving the lives of individuals living with spinal cord injuries worldwide.