The World Health Organization (WHO) has published today updated target regimen profiles for tuberculosis treatment and new target product profiles for tests for tuberculosis treatment monitoring and optimization. These documents define the most important characteristics and requirements to be considered for developing new tuberculosis (TB) treatment regimens and future tests to choose the best-suited TB treatments and monitor their effect.
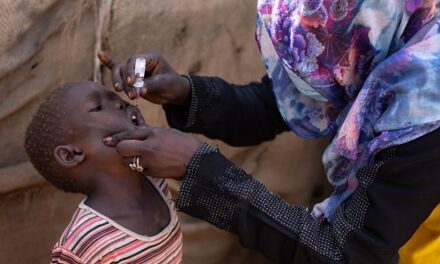
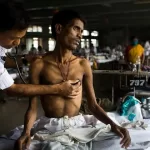
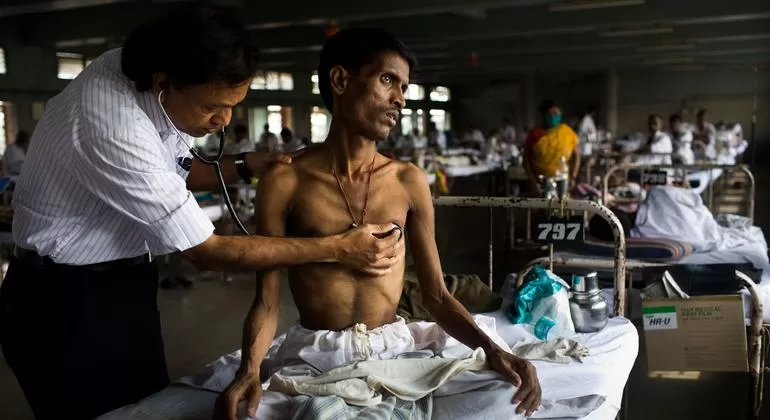
TB treatment regimens are lengthy and can be associated with both adverse effects and significant costs, which impact adherence and treatment outcomes. Treatment success for drug-susceptible TB has stagnated at around 86% globally for several years. In people treated with second-line regimens for multidrug- or rifampicin-resistant TB, the treatment success is only 60% and has only increased slightly over the last decade. The number of people with TB who die, sustain treatment failure, relapse or abandon treatment remains unacceptably high.
There is, therefore, an urgent need for simpler, shorter, safer, more tolerable, and more effective treatment regimens for all forms of tuberculosis that are easily accessible to all patients in need. In addition, better tests to choose and monitor treatment could improve overall treatment success. A better test is needed for a more reliable identification of relapse-free cure in people who have received TB treatment.
The target regimen profiles for tuberculosis treatment contain targets for treatment regimens for rifampicin-susceptible TB and rifampicin-resistant TB, outlining areas where progress is most urgently needed. In addition, the desired characteristics for a pan-TB treatment regimen as a better first-line regimen for all is described. The document is an update.
The target product profiles for tests for tuberculosis treatment monitoring and optimization contain targets for tests for treatment monitoring and cure and targets for tests that, in the future, might help TB treatment stratification by identifying patients who require a more intensive treatment regimen which may be longer or contain more medicines. This is the first document published by WHO on this topic.
“Together, these documents provide a clear direction for funders, developers, and researchers about characteristics required for new TB treatments and tests which are essential to ensure people-centered care and reach the global End TB targets,” said Tereza Kasaeva, Director of WHO’s Global Tuberculosis Programme. “WHO thanks the broad constituency of experts, TB survivors and other partners who contributed to their development.”