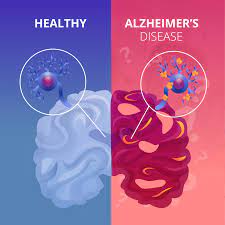
ROCHESTER, Minn. — In a discovery that could redefine our understanding of why dementia disproportionately affects biological females, a groundbreaking Mayo Clinic study has identified a “turbocharged” link between a protein associated with Parkinson’s disease and the rapid progression of Alzheimer’s pathology.
The research, published March 3, 2026, in JAMA Network Open, reveals that alpha-synuclein—the toxic protein responsible for Lewy body dementia and Parkinson’s—acts as a potent catalyst for Alzheimer’s-related brain changes specifically in women. While the protein’s presence has long been noted in advanced dementia cases, the Mayo study found it accelerates the accumulation of tau “tangles” in women at a rate up to 20 times faster than in men with identical protein profiles.
With women already accounting for nearly two-thirds of the 6.9 million Alzheimer’s cases in the United States, these findings provide a critical biological clue to a long-standing medical mystery: why the disease often hits women harder and faster.
A Toxic Duo: How Proteins Collide
In a healthy brain, proteins like tau and alpha-synuclein perform routine cellular functions. However, in neurodegenerative diseases, these proteins misfold and clump together, becoming toxic to neurons.
In Alzheimer’s disease, tau forms “tangles” that choke off a neuron’s internal transport system. In Parkinson’s, alpha-synuclein forms “Lewy bodies” that disrupt dopamine production and cognitive function. When both proteins coexist—a condition known as co-pathology—the results are devastating.
The Mayo Clinic team analyzed data from 415 participants through the Alzheimer’s Disease Neuroimaging Initiative (ADNI). By using advanced cerebrospinal fluid (CSF) testing and tau-PET brain imaging, researchers tracked how these proteins interacted over an average of 2.5 years.
“We found that in women who possessed both tau and alpha-synuclein abnormalities, the tau spread wasn’t just slightly faster—it was explosive,” said lead author Elijah Mak, Ph.D., a neuroimaging researcher at Mayo Clinic. “This suggests that alpha-synuclein may ‘seed’ or ignite the spread of tau, particularly in the female brain.”
The Sex Disparity: Why Women?
The study’s most striking finding was the sex-specific nature of this acceleration. While men in the study also showed presence of the Parkinson’s protein, they did not experience the same rapid “runaway” tau accumulation seen in their female counterparts.
Experts suggest several biological mechanisms could be at play:
-
Hormonal Shifts: The loss of estrogen during menopause may impair the brain’s ability to clear toxic proteins.
-
Genetic Factors: Interactions with the APOE4 gene, the strongest genetic risk factor for late-onset Alzheimer’s, are often more pronounced in women.
-
Immune Response: Differences in how female microglia (the brain’s immune cells) respond to protein clumps may inadvertently speed up damage.
“When we observe disease-related alterations developing at significantly different rates, we cannot continue treating Alzheimer’s as if it behaves uniformly across all patients,” noted senior author Kejal Kantarci, M.D., a Mayo Clinic neuroradiologist.
Expert Perspectives: A Call for Precision Medicine
Independent experts not involved in the study emphasize that this research marks a shift toward “precision neurology”—treating patients based on their specific biological markers rather than a one-size-fits-all diagnosis.
“The 20-fold difference reported here is striking,” said Dr. Joanna Hellier, a neurologist at Johns Hopkins University. “It highlights a critical need to screen for Parkinson’s proteins even in patients who only show traditional Alzheimer’s symptoms. If we are only looking at one protein, we are only seeing half the fire.”
Dr. Maria Carrillo, Chief Science Officer at the Alzheimer’s Association, added that this underscores the necessity for sex-stratified research. “Targeting alpha-synuclein alongside amyloid and tau could finally address why certain therapies have historically succeeded less in women than in men.”
Public Health: What This Means for You
For the general public, this study shifts the conversation from “prevention” to “early detection and personalized management.”
As of 2025, several blood-based biomarkers and tau-PET scans have received FDA approval, making it easier for clinicians to identify high-risk protein profiles before significant cognitive decline occurs.
Practical Steps for Health-Conscious Consumers:
-
Discuss Family History: If you have a family history of both Parkinson’s and Alzheimer’s, mention this to your primary care physician or neurologist.
-
Monitor “Mixed” Symptoms: Be aware that “mixed dementia”—showing symptoms of both memory loss and motor changes (like tremors or stiffness)—may indicate the presence of co-pathology.
-
Lifestyle Intervention: While research into specific drugs continues, the “pillars of brain health”—a Mediterranean diet, regular aerobic exercise, and cognitive training—remain the most effective ways to build “cognitive reserve” and potentially slow protein accumulation.
Limitations and the Road Ahead
While the results are groundbreaking, researchers urge caution. The study was observational, meaning it identifies a strong link but does not yet prove that alpha-synuclein causes the tau spread.
Furthermore, the ADNI cohort consists primarily of well-educated, white participants. “To truly understand this 20-fold acceleration, we must see if it holds true across diverse populations, including Black and Hispanic women who are statistically at even higher risk for dementia,” Dr. Hellier noted.
Future clinical trials may soon begin testing alpha-synuclein inhibitors—drugs currently being developed for Parkinson’s—in Alzheimer’s patients who test positive for the “synergy” of proteins.
Conclusion
The Mayo Clinic study serves as a wake-up call for the medical community. By identifying alpha-synuclein as a “fast-forward button” for Alzheimer’s in women, researchers have opened a new door for targeted therapies that could one day halt the progression of the world’s most common form of dementia.
References
-
Study Citation: Mak, E., et al. (2026). Sex-Specific Associations of α-Synuclein Pathology With Tau Accumulation Across the Alzheimer Disease Continuum. JAMA Network Open. doi:10.1001/jamanetworkopen.2026.0461
Medical Disclaimer: This article is for informational purposes only and should not be considered medical advice. Always consult with qualified healthcare professionals before making any health-related decisions or changes to your treatment plan. The information presented here is based on current research and expert opinions, which may evolve as new evidence emerges.